Physics:Quantum atoms/spin
Jump to navigation
Jump to search
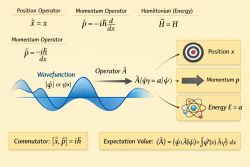
Spin is an intrinsic form of angular momentum carried by an electron and other quantum particles. It is a fundamental property that does not correspond to classical rotation.
Spin states of an electron, often described as "up" or "down".
Description
Spin is quantized and can take discrete values. For electrons, spin has two possible states, commonly referred to as "spin-up" and "spin-down". These states play a central role in determining how electrons occupy orbitals.
Spin is essential for understanding atomic structure, magnetic properties, and the Pauli exclusion principle.
Properties
- intrinsic quantum property
- related to angular momentum
- determines electron pairing in atoms
See also
Table of contents (185 articles)
Index
Core theory
Applications and extensions
Full contents
1. Foundations (11) ↑ Back to index
2. Conceptual and interpretations (14) ↑ Back to index
3. Mathematical structure and systems (13) ↑ Back to index
4. Atomic and spectroscopy (14) ↑ Back to index
5. Wavefunctions and modes (9) ↑ Back to index
6. Quantum dynamics and evolution (17) ↑ Back to index
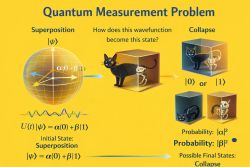
7. Measurement and information (9) ↑ Back to index
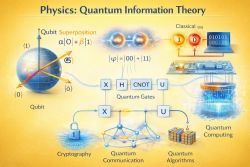
8. Quantum information and computing (10) ↑ Back to index
9. Quantum optics and experiments (5) ↑ Back to index
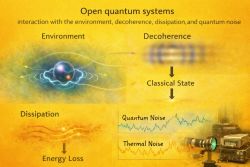
10. Open quantum systems (9) ↑ Back to index
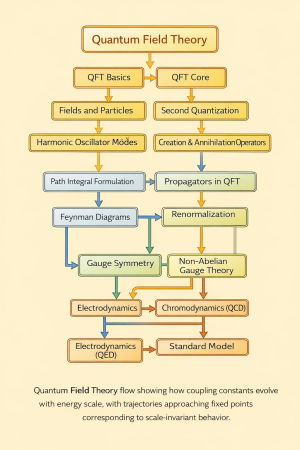
11. Quantum field theory (20) ↑ Back to index
12. Statistical mechanics and kinetic theory (9) ↑ Back to index
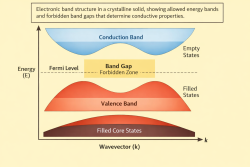
13. Condensed matter and solid-state physics (13) ↑ Back to index
149. Physics:Quantum well
154. Physics:Quantum dot
14. Plasma and fusion physics (8) ↑ Back to index
15. Timeline (8) ↑ Back to index
16. Advanced and frontier topics (16) ↑ Back to index
References
Source attribution: Physics:Quantum atoms/spin