Physics:Quantum Hyperfine structure
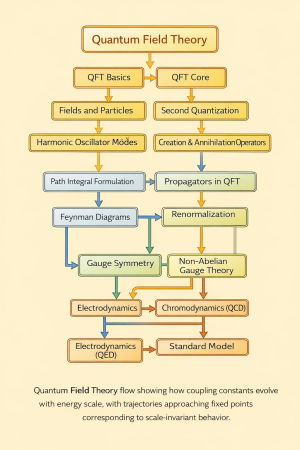
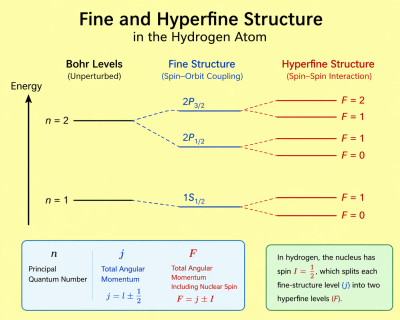
Hyperfine structure refers to small shifts and splittings of otherwise degenerate electronic energy levels in atoms, molecules, and ions due to interactions between the nucleus and the surrounding electron cloud.
These shifts are typically much smaller than those of fine structure and arise from electromagnetic multipole interactions, primarily involving nuclear magnetic dipole and electric quadrupole moments.
Overview
In atomic systems, hyperfine structure originates from:
- interaction between the nuclear magnetic dipole moment and magnetic fields produced by electrons
- interaction between the nuclear electric quadrupole moment and the electric field gradient
In molecules, additional contributions arise from:
- nuclear spin–spin interactions
- nuclear spin–rotation coupling
Hyperfine structure is fundamentally weaker than fine structure and reflects the coupling between nuclear and electronic degrees of freedom.
Magnetic dipole interaction
For a nucleus with spin , the magnetic dipole moment is
where is the nuclear g-factor and the nuclear magneton.
The interaction Hamiltonian is:
where is the magnetic field generated by electrons.
In the effective angular momentum form, this becomes:
leading to the hyperfine energy shift:
where:
- = total electronic angular momentum
- = total angular momentum
This interaction satisfies the Landé interval rule.
Electric quadrupole interaction
For nuclei with spin , an electric quadrupole moment exists.
The quadrupole Hamiltonian is:
where:
- describes the nuclear quadrupole moment
- describes the electric field gradient
This interaction reflects deviations from spherical nuclear charge distributions.
Molecular hyperfine structure
In molecules, hyperfine structure includes additional contributions:
Spin–spin interaction
Magnetic coupling between nuclei:
Spin–rotation interaction
Coupling between nuclear spins and molecular rotation:
These effects are important in rotational spectroscopy.
Experimental observation
Hyperfine structure is observed in:
- atomic spectra
- molecular spectroscopy
- electron paramagnetic resonance
- nuclear magnetic resonance
A key example is the 21 cm hydrogen line in astrophysics.
Applications
Atomic clocks
The SI second is defined via the hyperfine transition of caesium-133:
One second equals exactly:
9192631770 cycles of this transition.
Astrophysics
Hyperfine transitions probe the interstellar medium and molecular clouds.
Quantum computing
Hyperfine states serve as long-lived qubits in trapped-ion systems.
Precision physics
Measurements of hyperfine splitting provide tests of quantum electrodynamics.
History
Hyperfine structure was first described theoretically by Enrico Fermi in 1930.[1]
The nuclear quadrupole moment was introduced in 1935 by H. Schüler and T. Schmidt.[2]
See also
Table of contents (185 articles)
Index
Full contents
References
Source attribution: Physics:Quantum Hyperfine structure