Physics:Quantum Hydrogen atom
The hydrogen atom is the simplest atomic system, consisting of a single electron bound to a proton by the Coulomb interaction. It is the only atom in quantum mechanics that admits a fully exact analytical solution of the Schrödinger equation, making it a fundamental model for understanding atomic structure, spectroscopy, and quantum theory.[1]
Schrödinger equation and Coulomb potential
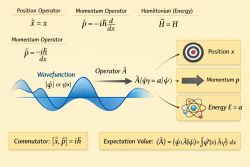
The electron in a hydrogen atom is described by the time-independent Schrödinger equation in a central Coulomb potential:
Because the potential depends only on the radial coordinate , the equation is separable in spherical coordinates.[2]
---
Quantum numbers
The solutions are characterized by three quantum numbers:
- Principal quantum number:
- Orbital angular momentum:
- Magnetic quantum number:
These arise from the separation of variables into radial and angular parts.[3]
---
Energy levels
The allowed energy levels depend only on the principal quantum number:
This degeneracy is a consequence of the underlying symmetry of the Coulomb potential.[4]
---
Wavefunctions and orbitals
The hydrogen wavefunctions are products of radial functions and spherical harmonics:
These define the familiar atomic orbitals:
- -orbitals () — spherical symmetry
- -orbitals () — directional lobes
- -orbitals () — more complex structures
---
Angular momentum
The orbital angular momentum is quantized:
The hydrogen atom also includes electron spin, introducing total angular momentum when relativistic effects are considered.[6]
---
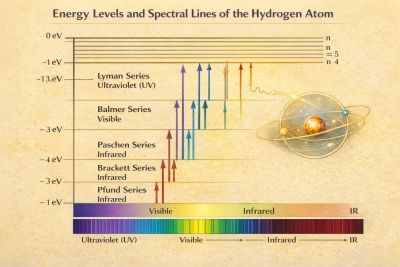
Spectral lines
Transitions between energy levels produce photons with energy:
This gives rise to discrete spectral series:
- Lyman series () — ultraviolet
- Balmer series () — visible
- Paschen, Brackett, Pfund — infrared
The wavelengths satisfy the Rydberg formula:
where is the Rydberg constant.[7][8]
---
Fine and hyperfine structure
More accurate treatments include:
- Fine structure — relativistic corrections and spin–orbit coupling
- Hyperfine structure — interaction between electron and nuclear spin
These effects lift degeneracies and produce small spectral splittings.[9][10]
---
Importance in quantum mechanics
The hydrogen atom plays a central role because:
- It provides an exact solution of the Schrödinger equation
- It explains atomic spectra quantitatively
- It reveals hidden symmetries (e.g., Runge–Lenz vector)
- It serves as the starting point for multi-electron approximations
---
See also
Table of contents (185 articles)
Index
Full contents
References
- ↑ The Hydrogen Atom (OpenStax/LibreTexts)
- ↑ Schrödinger Theory of Hydrogen
- ↑ Hydrogen Atomic Orbitals Depend upon Three Quantum Numbers
- ↑ Atomic Data for Hydrogen (NIST)
- ↑ The Schrödinger Wave Equation for the Hydrogen Atom
- ↑ The Hydrogen Atom (OpenStax/LibreTexts)
- ↑ Bohr's Theory of the Hydrogen Atom (OpenStax)
- ↑ NIST Fundamental Physical Constants
- ↑ Fine Structure of Hydrogen
- ↑ Hyperfine Structure
Source attribution: Quantum Hydrogen atom