Physics:Quantum system
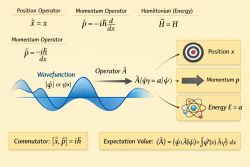
A quantum system is a physical system whose state, properties, and possible measurement outcomes are described by quantum mechanics. In quantum theory, physical states are represented in a mathematical state space, usually a Hilbert space, and observables are represented by operators.[1] A quantum system may be as small as a single electron, atom, or molecule, or as large as a many-particle system such as a crystal, gas, plasma, or quantum field.
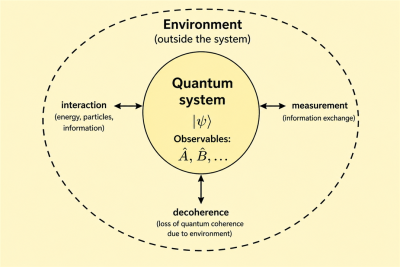
In quantum mechanics, the word system refers to the part of the physical world chosen for study. Everything outside that chosen part is treated as its surroundings or environment. The boundary between system and environment is not fixed by nature; it is a modeling choice made according to the physical question being asked.[2]
Quantum state
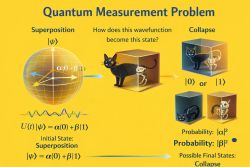
The condition of a quantum system is described by a quantum state. For a simple isolated system, this state may be represented by a wave function or by a state vector in Hilbert space. For systems with uncertainty, mixing, or interaction with an environment, the state may be represented more generally by a density operator.[2]
The quantum state is used to calculate probabilities for measurement outcomes. These outcomes may involve quantities such as position, momentum, energy, spin, angular momentum, or particle number. Unlike in classical physics, a quantum state does not always assign definite pre-existing values to all observable properties.[1]
Observables and measurement
Physical quantities that can be measured in a quantum system are called observables. In the mathematical formulation of quantum mechanics, observables are represented by operators.[3] A measurement can change the state of the system, especially when the measured quantity is not already definite in that state.
This feature makes measurement central to quantum theory. The same quantum system can give different outcomes in repeated measurements, but the probabilities of those outcomes are determined by its quantum state.
System and environment
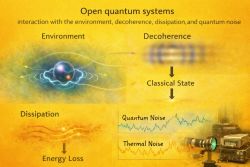
A quantum system is often treated as isolated in simple models, but real systems usually interact with their surroundings. These interactions can exchange energy, particles, or information with the environment. A system that interacts with an external environment is called an open quantum system.[2]
Interaction with the environment may also cause loss of quantum coherence, a process known as decoherence.[4]
The system-environment division is especially important in quantum measurement, open quantum systems, quantum information, and condensed-matter physics. In each case, the chosen boundary determines which degrees of freedom are described directly and which are treated as external influences.
Examples
Examples of quantum systems include:
- a single electron in an electromagnetic field;
- an atom with discrete energy levels;
- a molecule described by molecular orbitals and vibrations;
- a photon in an optical cavity;
- a spin system in a magnetic field;
- a superconductor or other many-body quantum material;
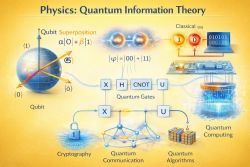
- a qubit used in quantum information and computing.
Classical and quantum description
Classical physics often describes a system by definite values of position, velocity, energy, and other properties. Quantum mechanics instead describes the system by a state that gives probability amplitudes or probabilities for possible measurement results.[1]
Quantum systems can display effects with no direct classical analogue, including superposition, interference, entanglement, quantized energy levels, and wave-particle duality. These effects are most visible at atomic, molecular, and subatomic scales, but they also underlie macroscopic quantum phenomena such as superconductivity and superfluidity.
See also
Table of contents (185 articles)
Index
Full contents
References
- ↑ 1.0 1.1 1.2 Template:Cite encyclopedia
- ↑ 2.0 2.1 2.2 Breuer, Heinz-Peter; Petruccione, Francesco (2002). The Theory of Open Quantum Systems. Oxford University Press. ISBN 978-0-19-852063-4.
- ↑ Landsman, N. P.. Lecture Notes on Hilbert Spaces and Quantum Mechanics. Radboud University Nijmegen. https://www.math.ru.nl/~landsman/HSQM.pdf. Retrieved 5 May 2026.
- ↑ Template:Cite encyclopedia
Source attribution: Quantum system