Physics:Quantum molecular structure
Quantum molecular structure describes the arrangement of atoms within molecules and the quantum-mechanical principles that determine molecular geometry, chemical bonding, molecular orbitals, and the structural properties of matter at the molecular scale. Molecular structure emerges from the interactions between atomic nuclei and electrons, governed by the laws of quantum mechanics and quantum chemistry.
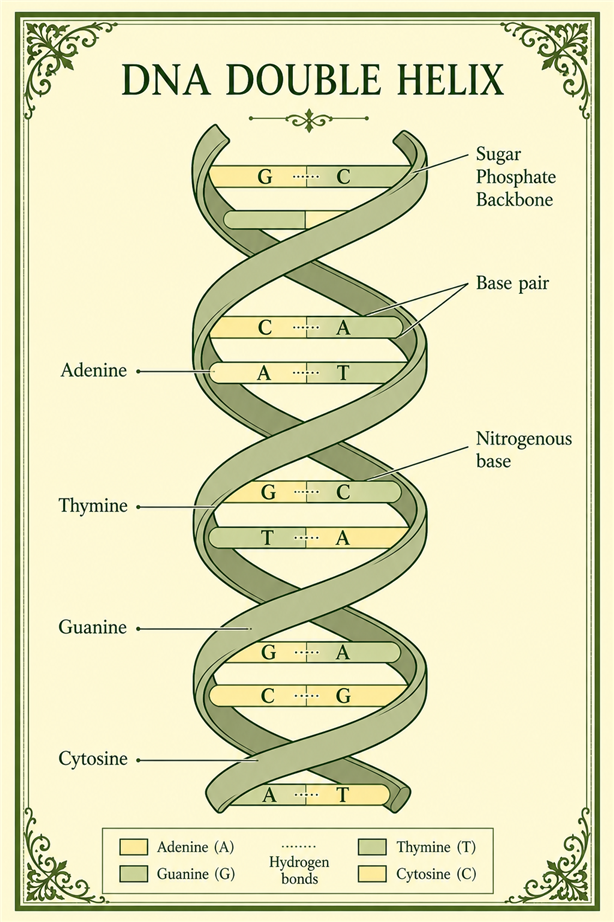
Diagrammatic representation of the structural features of the DNA double helix. Molecular geometry and hydrogen bonding determine the organization and replication properties of DNA.
Overview
Molecular structure concerns the spatial arrangement of atoms and the distribution of electrons within molecules. These structures determine many observable physical and chemical properties, including stability, reactivity, optical behavior, electrical properties, and biological function.[1]
Quantum mechanics explains molecular structure through the behavior of electrons occupying quantized molecular orbitals. Electrons interact through electromagnetic forces and occupy allowed energy states determined by the Schrödinger equation.[2]
The geometry of molecules is influenced by:
- electron configuration
- orbital hybridization
- electrostatic interactions
- molecular symmetry
- quantum exchange effects
- hydrogen bonding
- intermolecular forces
Chemical bonding
Chemical bonds arise from electromagnetic interactions between atoms and the quantum-mechanical sharing or transfer of electrons.[3]
Major bonding types include:
- covalent bonds
- ionic bonds
- metallic bonding
- van der Waals interactions
- hydrogen bonding
Quantum mechanics explains why electrons occupy discrete orbitals and why certain molecular configurations are energetically favorable.[4]
Molecular orbitals
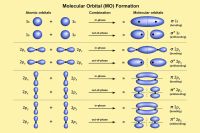
In molecular orbital theory, atomic orbitals combine to form molecular orbitals extending over the entire molecule.[5]
Electrons occupy bonding, antibonding, or nonbonding orbitals depending on energy and symmetry considerations. Molecular orbital theory explains:
- molecular stability
- bond order
- electronic transitions
- spectroscopy
- conductivity
- magnetic properties
Molecular geometry
Molecular geometry describes the three-dimensional arrangement of atoms. The shape of molecules depends on the distribution of electrons and the minimization of energy.[6]
Common molecular geometries include:
- linear
- trigonal planar
- tetrahedral
- trigonal bipyramidal
- octahedral
These structures strongly influence molecular interactions and physical properties.
Spectroscopy and diffraction
Experimental methods used to determine molecular structure include:
- spectroscopy
- X-ray diffraction
- neutron diffraction
- electron diffraction
- nuclear magnetic resonance
- infrared spectroscopy
- Raman spectroscopy
X-ray crystallography became one of the most important methods for determining complex molecular structures.[7]
DNA double helix
One of the most important discoveries in molecular structure was the determination of the DNA double helix.[8]
The structure was determined using X-ray diffraction experiments together with quantum and chemical models of molecular bonding.[9][10]
The DNA molecule consists of two complementary strands held together by hydrogen bonds between nucleotide base pairs. The helical structure explains how genetic information can be stored and replicated.[11]
The discovery of DNA structure transformed molecular biology and demonstrated how quantum-scale interactions could produce highly organized biological systems.
Physical molecular templates used by Watson and Crick during construction of the DNA double-helix model.
Molecular biology and quantum science
The development of molecular structure theory contributed directly to:
- molecular biology
- biochemistry
- nanotechnology
- biotechnology
- materials science
- pharmaceutical chemistry
- quantum chemistry
Understanding molecular structure made possible the interpretation of the genetic code, protein folding, enzyme function, and many biological processes.[12]
See also
Table of contents (72 articles)
Index
Full contents
References
- ↑ Pauling, Linus (1960). The Nature of the Chemical Bond. Cornell University Press.
- ↑ Atkins, Peter (2018). Physical Chemistry. Oxford University Press.
- ↑ Levine, Ira N. (2014). Quantum Chemistry. Pearson.
- ↑ Pauling, Linus (1931). "The Nature of the Chemical Bond". Journal of the American Chemical Society 53 (4): 1367–1400. doi:10.1021/ja01355a027.
- ↑ Mulliken, Robert S. (1932). "Electronic Structures of Polyatomic Molecules and Valence". Physical Review 41 (1): 49–71. doi:10.1103/PhysRev.41.49.
- ↑ Housecroft, Catherine (2018). Inorganic Chemistry. Pearson.
- ↑ Bragg, William Lawrence (1914). "The Diffraction of Short Electromagnetic Waves by a Crystal". Proceedings of the Cambridge Philosophical Society 17: 43–57.
- ↑ "Molecular structure of nucleic acids; a structure for deoxyribose nucleic acid". Nature 171 (4356): 737–738. April 1953. doi:10.1038/171737a0. PMID 13054692. Bibcode: 1953Natur.171..737W. https://www.nature.com/articles/171737a0.
- ↑ "Molecular configuration in sodium thymonucleate". Nature 171 (4356): 740–741. 1953-04-25. doi:10.1038/171740a0. PMID 13054694. Bibcode: 1953Natur.171..740F. http://www.nature.com/nature/dna50/franklingosling.pdf.
- ↑ "Molecular structure of deoxypentose nucleic acids". Nature 171 (4356): 738–740. 25 April 1953. doi:10.1038/171738a0. PMID 13054693. Bibcode: 1953Natur.171..738W. http://www.nature.com/nature/dna50/wilkins.pdf.
- ↑ Perutz, MF (June 1969). "DNA helix". Science 164 (3887): 1537–1539. doi:10.1126/science.164.3887.1537. PMID 5796048. Bibcode: 1969Sci...164.1537W.
- ↑ Judson, Horace Freeland (1979). The Eighth Day of Creation: Makers of the Revolution in Biology. Simon & Schuster. ISBN 9780671254100.
Source attribution: Molecular Structure of Nucleic Acids: A Structure for Deoxyribose Nucleic Acid