Physics:Quantum atoms/transition
A transition is a change of an electron between different energy levels in an atom. Such transitions occur when energy is absorbed or emitted, typically in the form of a photon.

Description
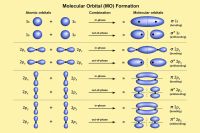
In quantum mechanics, electrons in atoms occupy discrete quantized energy levels. An atomic electron transition (also called a quantum jump or quantum leap) occurs when an electron changes from one energy level to another within an atom or artificial atom.[6][7]
These energy levels are unique to each atom and produce characteristic spectral fingerprints. Techniques such as energy-dispersive X-ray spectroscopy and X-ray photoelectron spectroscopy rely on these characteristic transitions to identify atomic composition.[8]
When an electron moves to a higher energy level, it absorbs energy. When it falls to a lower level, it emits energy. These processes are governed by quantum-mechanical selection rules and conservation of energy.
Transitions between energy levels produce discrete spectral features and are fundamental to atomic spectroscopy.
Photon absorption and emission
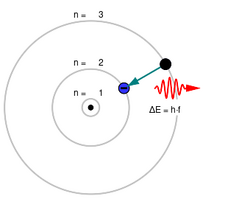
Electrons can relax into lower-energy states by emitting electromagnetic radiation in the form of photons. Conversely, they can absorb photons and become excited into higher-energy states.
The energy of the photon must exactly match the energy difference between the two states. Larger energy gaps correspond to shorter photon wavelengths.[9]
The relation between photon energy and frequency is:
- contentReference[oaicite:0]{index=0}
where Template:Math is the Planck constant, Template:Math is frequency, Template:Math is the speed of light, and Template:Math is wavelength.
Quantum theory
An atom interacting with electromagnetic radiation experiences an oscillating electric field:
- contentReference[oaicite:1]{index=1}
where Template:Math is the angular frequency and Template:Math is the polarization vector.[10]
The interaction Hamiltonian for an atomic dipole in an electric field is:
- contentReference[oaicite:2]{index=2}
Using time-dependent perturbation theory and Fermi’s golden rule, the stimulated transition probability depends on the dipole matrix element:
- contentReference[oaicite:3]{index=3}
The angular part of this expression leads directly to the quantum-mechanical selection rules for atomic transitions.
Electromagnetic radiation interactions
To excite an electron into a higher energy level, incident radiation must have energy equal to the energy gap between the levels. Because atomic energy differences are often on the scale of ultraviolet and X-ray photons, these wavelengths are widely used in spectroscopy.[8]
The Franck–Condon principle states that electronic transitions occur much faster than nuclear motion. As a result, transitions occur essentially instantaneously compared to atomic vibrations and are only likely if the initial and final wavefunctions overlap significantly.[11]
Radiative relaxation produces photons with wavelengths characteristic of the atom and transition involved.
Spectroscopy techniques
Several experimental methods use electron transitions:
- Ultraviolet–visible spectroscopy uses visible or ultraviolet light to probe absorption and transmission spectra.[12]
- Energy-dispersive X-ray spectroscopy excites inner-shell electrons using high-energy electrons and measures emitted X-rays characteristic of the atom.[13]
- X-ray photoelectron spectroscopy uses incident X-rays to eject electrons from surfaces and determine elemental composition from their binding energies.[14]
History
Danish physicist Niels Bohr first proposed quantum jumps in 1913.[15] Shortly afterward, the Franck–Hertz experiment by James Franck and Gustav Hertz experimentally confirmed that atoms possess quantized energy states.[16]
In 1975, Hans Dehmelt predicted that individual quantum jumps could be observed directly. In 1986, quantum jumps were experimentally observed using trapped ions of barium and mercury.[9]
Recent discoveries
In 2019, experiments with superconducting artificial atoms demonstrated that some quantum jumps evolve continuously and can even be reversed during the transition.[17]
Other quantum jumps remain fundamentally unpredictable due to the probabilistic nature of quantum measurement.[18]
Properties
- involves energy levels
- associated with emission or absorption of photons
- produces spectral lines
- governed by quantum selection rules
- fundamental to spectroscopy and laser physics
See also
Table of contents (72 articles)
Index
Full contents
References
- ↑ Schombert, James. "Quantum physics". University of Oregon Department of Physics.
- ↑ McQuarrie, Donald A.; Simon, John D.. Physical chemistry: a molecular approach. Univ. Science Books.
- ↑ Itano, W. M.; Bergquist, J. C.; Wineland, D. J.. "Early observations of macroscopic quantum jumps in single atoms". International Journal of Mass Spectrometry 377: 403.
- ↑ Foot, C. J.. Atomic Physics. Oxford University Press.
- ↑ Template:Cite news
- ↑ Schombert, James. "Quantum physics" University of Oregon Department of Physics
- ↑ Vijay, R; Slichter, D. H; Siddiqi, I (2011). "Observation of Quantum Jumps in a Superconducting Artificial Atom". Physical Review Letters 106 (11). doi:10.1103/PhysRevLett.106.110502. PMID 21469850. Bibcode: 2011PhRvL.106k0502V.
- ↑ 8.0 8.1 McQuarrie, Donald A.; Simon, John D. (200). Physical chemistry: a molecular approach. Sausalito, Calif: Univ. Science Books. ISBN 978-0-935702-99-6.
- ↑ 9.0 9.1 Itano, W. M.; Bergquist, J. C.; Wineland, D. J. (2015). "Early observations of macroscopic quantum jumps in single atoms". International Journal of Mass Spectrometry 377: 403. doi:10.1016/j.ijms.2014.07.005. Bibcode: 2015IJMSp.377..403I. http://tf.boulder.nist.gov/general/pdf/2723.pdf.
- ↑ Foot, CJ (2004). Atomic Physics. Oxford University Press. ISBN 978-0-19-850696-6.
- ↑ de la Peña, L.; Cetto, A. M.; Valdés-Hernández, A. (2020-12-04). "How fast is a quantum jump?". Physics Letters A 384 (34). doi:10.1016/j.physleta.2020.126880. ISSN 0375-9601. Bibcode: 2020PhLA..38426880D. https://www.sciencedirect.com/science/article/pii/S0375960120307477.
- ↑ "UV-Visible Spectroscopy". https://www2.chemistry.msu.edu/faculty/reusch/virttxtjml/spectrpy/uv-vis/uvspec.htm.
- ↑ Template:Citation
- ↑ "X-ray Photoelectron Spectroscopy" (in en). https://serc.carleton.edu/msu_nanotech/methods/xps.html.
- ↑ Template:Cite news
- ↑ "Franck-Hertz experiment | physics | Britannica" (in en). https://www.britannica.com/science/Franck-Hertz-experiment.
- ↑ Minev, Z. K.; Mundhada, S. O.; Shankar, S.; Reinhold, P.; Gutiérrez-Jáuregui, R.; Schoelkopf, R. J..; Mirrahimi, M.; Carmichael, H. J. et al. (3 June 2019). "To catch and reverse a quantum jump mid-flight". Nature 570 (7760): 200–204. doi:10.1038/s41586-019-1287-z. PMID 31160725. Bibcode: 2019Natur.570..200M.
- ↑ Snizhko, Kyrylo; Kumar, Parveen; Romito, Alessandro (2020-09-29). "Quantum Zeno effect appears in stages". Physical Review Research 2 (3). doi:10.1103/PhysRevResearch.2.033512. Bibcode: 2020PhRvR...2c3512S. https://link.aps.org/doi/10.1103/PhysRevResearch.2.033512.
Source attribution: Physics:Quantum atoms/transition