Physics:Quantum photon
A quantum photon is the elementary quantum of the electromagnetic field. It is the particle-like unit of electromagnetic radiation, including visible light, radio waves, X-rays, and gamma rays. In the Standard Model, the photon is an elementary boson and the gauge boson of the electromagnetic interaction.[1]
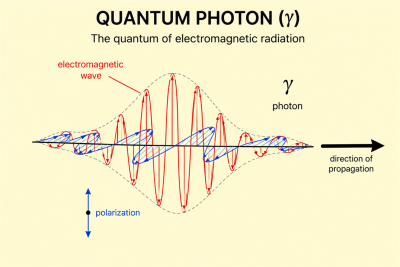
Photons are usually described as massless, electrically neutral, stable spin-1 particles. In vacuum they propagate at the speed of light and have two physical polarization states. A photon carries energy and momentum even though it has no rest mass, and its energy is proportional to its frequency.[2][3]
Quantum description
In quantum physics, a photon is not simply a tiny classical object. It is a quantum excitation of an electromagnetic mode. When light is emitted or absorbed, energy is transferred in discrete units called photons. The energy of one photon is
where is the Planck constant, is the frequency, is the reduced Planck constant, and is the angular frequency. In terms of wavelength,
where is the speed of light in vacuum and is the wavelength.[2]
The momentum of a photon is
- .
This relation is consistent with the relativistic energy–momentum relation for a particle with zero rest mass.
Bosonic nature
A photon is a spin-1 boson. Because photons obey Bose–Einstein statistics, more than one photon can occupy one shared quantum state. This property is central to coherent light, lasers, black-body radiation, and the quantum theory of electromagnetic fields.[4][5]
Unlike fermions, photons do not obey the Pauli exclusion principle. This is why an electromagnetic mode can contain many photons, and why coherent optical fields can be built from large numbers of photons in related quantum states.
Wave–particle duality
Photons show wave–particle duality. In experiments such as interference and diffraction, photon probabilities follow wave-like patterns. Yet when a photon is detected, it is registered as a discrete event. A single photon can therefore produce an interference pattern only statistically, after many repeated detections.[6][7]
This dual behavior is one reason photons are important in the foundations of quantum mechanics. They connect the wave theory of light with the particle-like transfer of energy, momentum, and angular momentum.
Physical properties
The photon has zero electric charge and is normally treated as having zero rest mass. Experimental tests place extremely small upper limits on any possible photon mass, but no nonzero photon mass has been established.[8][9]
A real photon in vacuum has two polarization states. These may be described in terms of helicity, corresponding to spin angular momentum components and along the direction of propagation.[3] Polarization links the quantum description of photons to the classical polarization of electromagnetic waves.
Historical development
The modern photon concept developed from the study of black-body radiation, the photoelectric effect, and the particle-like behavior of light. Max Planck introduced quantized energy elements in his work on black-body radiation. Albert Einstein then proposed in 1905 that light itself could be treated as localized energy quanta in order to explain the photoelectric effect.[10][11]
The name photon was popularized by Gilbert N. Lewis in 1926 for the quantum of light.[12][13] Arthur Compton's X-ray scattering experiments later provided strong evidence that light quanta carry momentum.[14]
Photon as a gauge boson
In quantum field theory, the photon is the gauge boson of electromagnetism. It is associated with a U(1) gauge symmetry and appears as the quantum carrier of the electromagnetic interaction.[15]
Virtual photons appear in quantum electrodynamics as intermediate carriers of electromagnetic interactions. They are not directly observed as free particles, but they are part of the mathematical description used to calculate electromagnetic scattering, radiative corrections, and forces between charged particles.[16]
Emission and absorption
Photons are emitted or absorbed when quantum systems change energy states. Atoms, molecules, nuclei, and solids can emit photons during transitions from higher to lower energy levels. Conversely, absorption of a photon can raise a system to a higher energy state or trigger processes such as ionization, photochemistry, or electronic excitation.
Einstein's theory of spontaneous and stimulated emission provided a statistical description of radiation interacting with matter. Stimulated emission later became the physical basis of the laser.[17][18]
In matter
When light travels through transparent matter, its effective speed is reduced by interactions with the material. In a quantum description, photons may couple to excitations of matter to form quasiparticles such as polaritons. Photons can also be scattered, absorbed, or re-emitted by atoms, molecules, and solids.
Photon interactions with matter are central to spectroscopy, photochemistry, microscopy, solar radiation transport, optical communication, and quantum technologies. In many of these applications, the photon picture is essential because energy is exchanged in discrete units.
Quantum information and optics
Photons are widely used in quantum optics and quantum information. Their polarization, path, phase, arrival time, or photon number can encode quantum states. Single photons and entangled photon pairs are used in tests of quantum mechanics, quantum cryptography, optical communication, and photonic approaches to quantum computing.[19][20]
Single-photon experiments, photon antibunching, and beam-splitter tests show that light cannot always be explained as a classical electromagnetic wave. These experiments support the quantum view of photons as individual excitations of electromagnetic modes.[21][7]
See also
Table of contents (72 articles)
Index
Full contents
References
- ↑ Amsler, C. (2008). "Review of Particle Physics". Physics Letters B 667 (1): 1–134. doi:10.1016/j.physletb.2008.07.018. Bibcode: 2008PhLB..667....1A.
- ↑ 2.0 2.1 Hecht, Eugene (1998). Optics (3rd ed.). Addison-Wesley. ISBN 978-0-201-83887-9.
- ↑ 3.0 3.1 Schwartz, Matthew D. (2014). Quantum Field Theory and the Standard Model. Cambridge University Press. ISBN 978-1-107-03473-0.
- ↑ Bose, Satyendra Nath (1924). "Plancks Gesetz und Lichtquantenhypothese". Zeitschrift für Physik 26 (1): 178–181. doi:10.1007/BF01327326. Bibcode: 1924ZPhy...26..178B.
- ↑ Einstein, Albert (1924). "Quantentheorie des einatomigen idealen Gases". Sitzungsberichte der Preussischen Akademie der Wissenschaften 1924: 261–267.
- ↑ "Interference Fringes with Feeble Light". Proceedings of the Cambridge Philosophical Society 15: 114. 1909.
- ↑ 7.0 7.1 Grangier, P.; Roger, G.; Aspect, A. (1986). "Experimental Evidence for a Photon Anticorrelation Effect on a Beam Splitter: A New Light on Single-Photon Interferences". Europhysics Letters 1 (4): 173–179. doi:10.1209/0295-5075/1/4/004. Bibcode: 1986EL......1..173G.
- ↑ Tu, Liang-Cheng; Luo, Jun; Gillies, George T. (2005). "The mass of the photon". Reports on Progress in Physics 68 (1): 77–130. doi:10.1088/0034-4885/68/1/R02. Bibcode: 2005RPPh...68...77T.
- ↑ Goldhaber, Alfred Scharff; Nieto, Michael Martin (2010). "Photon and graviton mass limits". Reviews of Modern Physics 82 (1): 939–979. doi:10.1103/RevModPhys.82.939. Bibcode: 2010RvMP...82..939G.
- ↑ Planck, Max (1901). "Über das Gesetz der Energieverteilung im Normalspectrum". Annalen der Physik 4 (3): 553–563. doi:10.1002/andp.19013090310. Bibcode: 1901AnP...309..553P.
- ↑ Einstein, Albert (1905). "Über einen die Erzeugung und Verwandlung des Lichtes betreffenden heuristischen Gesichtspunkt". Annalen der Physik 17 (6): 132–148. doi:10.1002/andp.19053220607. Bibcode: 1905AnP...322..132E.
- ↑ Lewis, Gilbert N. (18 December 1926). "The conservation of photons". Nature 118 (2981): 874–875. doi:10.1038/118874a0. Bibcode: 1926Natur.118..874L.
- ↑ Template:Cite arXiv
- ↑ Compton, Arthur H. (1923). "A quantum theory of the scattering of X-rays by light elements". Physical Review 21 (5): 483–502. doi:10.1103/PhysRev.21.483. Bibcode: 1923PhRv...21..483C.
- ↑ Ryder, L. H. (1996). Quantum Field Theory (2nd ed.). Cambridge University Press. ISBN 978-0-521-47814-4.
- ↑ Feynman, Richard P. (1985). QED: The Strange Theory of Light and Matter. Princeton University Press. ISBN 978-0-691-12575-6.
- ↑ Einstein, Albert (1916). "Strahlungs-emission und -absorption nach der Quantentheorie". Verhandlungen der Deutschen Physikalischen Gesellschaft 18: 318–323. Bibcode: 1916DPhyG..18..318E.
- ↑ Dirac, Paul A. M. (1927). "The Quantum Theory of the Emission and Absorption of Radiation". Proceedings of the Royal Society A 114 (767): 243–265. doi:10.1098/rspa.1927.0039. Bibcode: 1927RSPSA.114..243D.
- ↑ Scully, M. O.; Zubairy, M. S. (1997). Quantum Optics. Cambridge University Press. ISBN 978-0-521-43595-6.
- ↑ Fox, Mark (2006). Quantum Optics: An Introduction. Oxford University Press. ISBN 978-0-19-856673-1.
- ↑ Kimble, H. J.; Dagenais, M.; Mandel, L. (1977). "Photon Antibunching in Resonance Fluorescence". Physical Review Letters 39 (11): 691–695. doi:10.1103/PhysRevLett.39.691. Bibcode: 1977PhRvL..39..691.
Source attribution: Photon