Physics:Quantum atoms/orbital
An atomic orbital is a quantum-mechanical function describing the location, wave-like behavior, and probability distribution of an electron in an atom.[1] Orbitals are central to electron configuration, chemical bonding, and atomic spectra.
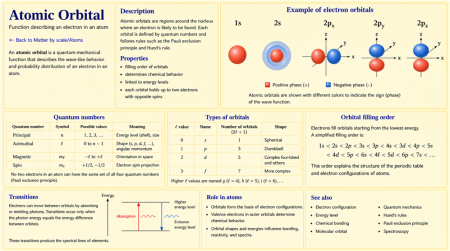
Description
In quantum mechanics, an orbital is not a fixed path followed by an electron. Instead, it is a wave function whose squared magnitude gives the probability density for finding the electron in a region around the nucleus.[2]
Atomic orbitals are characterized by quantum numbers. The principal quantum number Template:Mvar is related to the shell and energy scale. The azimuthal quantum number Template:Mvar determines the orbital type and angular momentum. The magnetic quantum number Template:Mvar describes orientation. A fourth quantum number, the spin projection Template:Mvar, describes electron spin.
An orbital can contain at most two electrons, and those two electrons must have opposite spin according to the Pauli exclusion principle.[3]
Electron properties
Electrons in atoms show both wave-like and particle-like behavior.
Wave-like properties include:
- electrons form standing-wave states around the nucleus
- the electron position is described probabilistically
- orbital shapes arise from wave interference and boundary conditions
Particle-like properties include:
- electrons carry a definite electric charge
- electrons occupy discrete quantum states
- transitions between orbitals involve absorption or emission of photons
This wave-particle behavior is why orbitals are often visualized as electron clouds rather than planetary orbits.
Quantum numbers
Atomic orbitals are labeled by quantum numbers:
- Template:Mvar — principal quantum number; shell, size, and energy scale
- Template:Mvar — azimuthal quantum number; orbital shape and angular momentum
- Template:Mvar — magnetic quantum number; orbital orientation
- Template:Mvar — spin quantum number; electron spin projection
The allowed values are restricted:
- Template:Mvar = 1, 2, 3, ...
- Template:Mvar = 0 to Template:Mvar − 1
- Template:Mvar = −Template:Mvar to +Template:Mvar
- Template:Mvar = +1/2 or −1/2
The Pauli exclusion principle states that no two electrons in the same atom can have the same set of all four quantum numbers.
Orbital names
Orbitals are named using the principal quantum number followed by a letter representing Template:Mvar:
- Template:Mvar = 0: s orbital
- Template:Mvar = 1: p orbital
- Template:Mvar = 2: d orbital
- Template:Mvar = 3: f orbital
The labels s, p, d, and f come from early spectroscopic descriptions: sharp, principal, diffuse, and fundamental. For Template:Mvar values above 3, the letters continue alphabetically as g, h, i, k, and so on.[4][5][6]
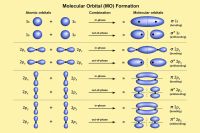
Shapes of orbitals
The simplest orbital is the s orbital, which is spherically symmetric. The p orbitals have two lobes and are oriented along different axes. The d and f orbitals have more complex lobed shapes.
Orbital images usually show surfaces enclosing regions where the electron has a high probability of being found. These diagrams do not show fixed paths; they show probability distributions.
The number and arrangement of nodes are determined by quantum numbers. Nodal surfaces are regions where the probability density is zero.
Hydrogen-like orbitals
The simplest exact orbitals are those of hydrogen-like atoms, which contain one electron. In such systems, the Schrödinger equation can be solved analytically.
Hydrogen-like orbitals are described using a radial part and an angular part:
For atoms with more than one electron, exact analytical solutions are not generally possible. Approximation methods such as Hartree–Fock theory and molecular orbital theory are used.
Orbital energy
In one-electron atoms, orbital energy depends mainly on Template:Mvar. In multi-electron atoms, electron-electron interactions make the energy depend also on Template:Mvar.
This explains why the filling order of orbitals in many-electron atoms is not simply 1s, 2s, 2p, 3s, 3p, 3d, and so on. Instead, the approximate filling sequence begins:
- 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p
This sequence is closely related to the structure of the periodic table and to electron configuration.
Electron configuration
Orbitals provide the framework for electron configuration. Electrons fill orbitals according to quantum rules such as:
- the Pauli exclusion principle
- Hund’s rule
- the Aufbau principle
The repeating structure of the periodic table arises because complete sets of s, p, d, and f orbitals can hold 2, 6, 10, and 14 electrons respectively.
Transitions between orbitals
Electrons can move between orbital states by absorbing or emitting photons. Such transitions occur only when the photon energy matches the energy difference between the two states.
These transitions produce spectral lines and are central to atomic spectroscopy.
History
Early atomic models treated electrons as orbiting particles. J. J. Thomson discovered the electron in 1897.[7] Hantaro Nagaoka proposed an orbit-based model in 1904.[8] Niels Bohr later introduced quantized electron orbits in 1913.[9]
The modern orbital concept emerged after the development of quantum mechanics, de Broglie matter waves, the Schrödinger equation, and Heisenberg’s uncertainty principle.[10][11]
The term orbital was introduced by Robert S. Mulliken in 1932 as a shortened form of one-electron orbital wave function.[12][13]
Properties
- describes electron probability density
- linked to energy levels
- determines chemical behavior
- provides the basis for electron configuration
- classified as s, p, d, f, and higher orbitals
- each orbital holds at most two electrons
- transitions between orbitals produce spectral lines
See also
Table of contents (72 articles)
Index
Full contents
References
- ↑ Orchin, Milton; MacOmber, Roger S.; Pinhas, Allan R.; Wilson, R. Marshall (2005). "Atomic Orbital Theory". The Vocabulary and Concepts of Organic Chemistry. pp. 1–24. doi:10.1002/0471713740.ch1. ISBN 978-0-471-68028-4.
- ↑ Daintith, J. (2004). Oxford Dictionary of Chemistry. New York: Oxford University Press. pp. 407–409. ISBN 978-0-19-860918-6. https://archive.org/details/dictionaryofchem0000unse_r3p4/page/408/mode/2up?view=theater.
- ↑ Levine, Ira N. (1991). Quantum Chemistry (4th ed.). Prentice-Hall. p. 262. ISBN 0-205-12770-3.
- ↑ Griffiths, David (1995). Introduction to Quantum Mechanics. Prentice Hall. pp. 190–191. ISBN 978-0-13-124405-4.
- ↑ Levine, Ira (2000). Quantum Chemistry (5 ed.). Prentice Hall. pp. 144–145. ISBN 978-0-13-685512-5. https://archive.org/details/quantumchemistry00levi_0/page/144.
- ↑ Atkins, Peter; de Paula, Julio; Friedman, Ronald (2009). Quanta, Matter, and Change: A Molecular Approach to Physical Chemistry. Oxford University Press. p. 106. ISBN 978-0-19-920606-3. https://books.google.com/books?id=QbQJAgAAQBAJ&pg=PA106.
- ↑ Thomson, J. J. (October 1897). "XL. Cathode Rays". The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science 44 (269): 293–316. doi:10.1080/14786449708621070.
- ↑ Nagaoka, H. (May 1904). "LV. Kinetics of a system of particles illustrating the line and the band spectrum and the phenomena of radioactivity". The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science 7 (41): 445–455. doi:10.1080/14786440409463141.
- ↑ Nicholson, J. W. (May 1914). "The Constitution of Atoms and Molecules". Nature 93 (2324): 268–269. doi:10.1038/093268a0. Bibcode: 1914Natur..93..268N.
- ↑ Heisenberg, W. (March 1927). "Über den anschaulichen Inhalt der quantentheoretischen Kinematik und Mechanik". Zeitschrift für Physik 43 (3–4): 172–198. doi:10.1007/BF01397280. Bibcode: 1927ZPhy...43..172H.
- ↑ Bohr, Niels (April 1928). "The Quantum Postulate and the Recent Development of Atomic Theory". Nature 121 (3050): 580–590. doi:10.1038/121580a0. Bibcode: 1928Natur.121..580B.
- ↑ Mulliken, Robert S. (July 1932). "Electronic Structures of Polyatomic Molecules and Valence. II. General Considerations". Physical Review 41 (1): 49–71. doi:10.1103/PhysRev.41.49. Bibcode: 1932PhRv...41...49M.
- ↑ Murrell, John N (5 September 2012). "The origins and later developments of molecular orbital theory". International Journal of Quantum Chemistry 112 (17): 2875–2879. doi:10.1002/qua.23293.
Source attribution: Atomic orbital