Physics:Quantum materials/band structure
Jump to navigation
Jump to search
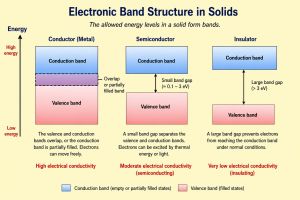
Band structure describes the allowed energy levels of electrons in a solid material. Instead of discrete energy levels, electrons occupy continuous ranges of energies called bands.
Electronic band structure determines whether a material behaves as a conductor, semiconductor, or insulator.
Description
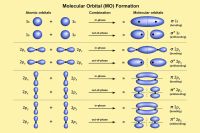
In a crystal lattice, the interaction between atoms causes individual electron energy levels to broaden into bands. Between these bands, there may be gaps where no electron states exist.
The arrangement of bands and gaps determines the electrical and optical properties of the material.
Properties
- continuous energy bands
- presence of band gaps
- determines conductivity
See also
Table of contents (72 articles)
Index
Composite matter
Sub-molecular
Full contents
1. Materials (6) ↑ Back to index
2. Matter (5) ↑ Back to index
3. Plasma and fusion physics (6) ↑ Back to index
4. Molecules (6) ↑ Back to index
5. Nuclear matter (6) ↑ Back to index
6. Atoms (7) ↑ Back to index
7. Particles (12) ↑ Back to index
8. Composite particles (12) ↑ Back to index
9. Fields (6) ↑ Back to index
10. Vacuum and spacetime (6) ↑ Back to index
References
Source attribution: Physics:Quantum materials/band structure