Physics:Quantum atoms/spectral line
Jump to navigation
Jump to search
A spectral line is a discrete frequency or wavelength of light emitted or absorbed by an atom. It arises from a transition of an electron between different energy levels.
File:Atomic spectrum lines.png
Spectral lines produced by electron transitions between discrete energy levels in an atom.
Description
Each spectral line corresponds to a specific difference in energy between two energy levels. When an electron transitions to a lower level, a photon is emitted; when it moves to a higher level, a photon is absorbed.
The set of spectral lines forms an atomic spectrum, which is characteristic of each element.
Properties
- discrete wavelengths or frequencies
- determined by energy level differences
- produced by transitions
See also
Table of contents (185 articles)
Index
Core theory
Applications and extensions
Full contents
1. Foundations (11) ↑ Back to index
2. Conceptual and interpretations (14) ↑ Back to index
3. Mathematical structure and systems (13) ↑ Back to index
4. Atomic and spectroscopy (14) ↑ Back to index
5. Wavefunctions and modes (9) ↑ Back to index
6. Quantum dynamics and evolution (17) ↑ Back to index
7. Measurement and information (9) ↑ Back to index
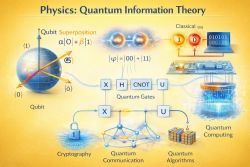
8. Quantum information and computing (10) ↑ Back to index
9. Quantum optics and experiments (5) ↑ Back to index
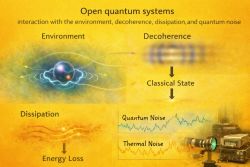
10. Open quantum systems (9) ↑ Back to index
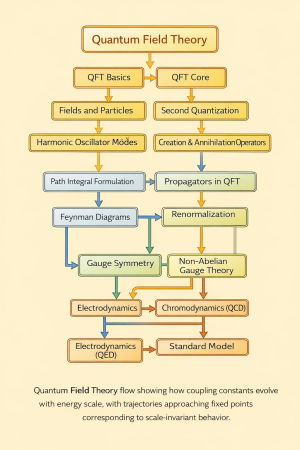
11. Quantum field theory (20) ↑ Back to index
12. Statistical mechanics and kinetic theory (9) ↑ Back to index
13. Condensed matter and solid-state physics (13) ↑ Back to index
149. Physics:Quantum well
154. Physics:Quantum dot
14. Plasma and fusion physics (8) ↑ Back to index
15. Timeline (8) ↑ Back to index
16. Advanced and frontier topics (16) ↑ Back to index
References
Source attribution: Physics:Quantum atoms/spectral line