Physics:Quantum mechanics/Timeline/Old quantum theory
The old quantum theory was a collection of ideas, rules, and semi-classical models developed between about 1900 and 1925, before the creation of modern quantum mechanics.[1] It was not a complete or self-consistent theory, but rather a set of heuristic corrections to classical mechanics.[2]
The old quantum theory introduced quantized energy, quantized angular momentum, atomic energy levels, and selection rules. Its central idea was that classical motion remained mostly valid, but only certain classical orbits or states were allowed. In modern terms, it is understood as a semi-classical approximation to quantum mechanics.[3][4]
The theory began with Max Planck's quantum hypothesis for black-body radiation and developed through Albert Einstein's work on light quanta and specific heat, Niels Bohr's model of the atom, and Arnold Sommerfeld's extensions of Bohr's model. It ended when Werner Heisenberg, Max Born, Pascual Jordan, and Erwin Schrödinger developed modern quantum mechanics in 1925–1926.
Historical background
The old quantum theory emerged from the failure of classical physics to explain several microscopic phenomena. Classical electrodynamics could not explain the stability of atoms, the discrete lines in atomic spectra, or the spectrum of black-body radiation. Classical statistical mechanics also failed to describe the low-temperature specific heat of solids.
Planck's 1900 work on black-body radiation introduced the quantum of action, now called the Planck constant.[5] Einstein then applied quantum ideas to the specific heat of solids in 1907, drawing attention to the physical significance of quantization.
The Einstein solid and later the Debye model applied quantum principles to atomic vibrations in solids and helped explain why the specific heat of solids decreases at low temperatures.
Early atomic models
Before Bohr, several physicists tried to apply quantum ideas to atomic structure. In 1910, Arthur Erich Haas developed a treatment of the hydrogen atom involving quantization of electronic orbits, anticipating parts of the later Bohr model.[6]
John William Nicholson was among the first to construct an atomic model using quantized angular momentum. In Nicholson's model, angular momentum was quantized in units of , a rule later central to the Bohr model.[7][8]
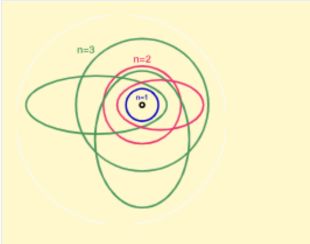
Bohr model
In 1913, Niels Bohr introduced a quantum model of the hydrogen atom. The model combined Rutherford's nuclear atom with Planck's quantum hypothesis. Bohr proposed that electrons could move only in certain allowed orbits and that radiation was emitted or absorbed when an electron jumped between these orbits.[9]
The model explained the hydrogen line spectrum and gave a theoretical foundation for the Rydberg formula. It also introduced a form of the correspondence principle, the idea that quantum theory must reproduce classical physics in the limit of large quantum numbers.
Although the Bohr model worked well for hydrogen-like atoms, it failed for more complex atoms and could not explain the intensities of spectral lines or the full structure of spectra.
Bohr–Sommerfeld theory
Arnold Sommerfeld extended Bohr's model by allowing elliptical orbits and by applying quantization rules to more general classical systems. The resulting Bohr–Sommerfeld model introduced additional quantum numbers and helped explain fine structure in atomic spectra.
Sommerfeld also quantized the z-component of angular momentum, a phenomenon then called space quantization.[10] This was an important step toward the later quantum-mechanical description of angular momentum.
The Bohr–Sommerfeld model was closer to the modern quantum-mechanical picture than Bohr's original circular orbit model, but it still relied on classical trajectories and could not provide a consistent microscopic theory.
Quantization condition
The main tool of the old quantum theory was the Bohr–Sommerfeld quantization condition. For a periodic classical motion, the action integral was required to be an integer multiple of Planck's constant:
where are momenta, are coordinates, and are integer quantum numbers.
The integral is an area in phase space and is called an action. This is why Planck's constant was often called the quantum of action. The rule worked only for systems whose classical motion was periodic or separable into periodic components.
This rule is also known as the Wilson–Sommerfeld quantization rule, since it was proposed independently by William Wilson and Arnold Sommerfeld.[11][12][13]
Specific heat and the harmonic oscillator
One of the earliest successes of quantum theory was the explanation of the low-temperature specific heat of solids. Classical thermodynamics predicted that each harmonic oscillator should contribute a constant amount of heat capacity. This disagreed with experiments, which showed that the specific heat of solids decreases toward zero at low temperature.
In the old quantum theory, the harmonic oscillator has quantized energy levels:
This quantization means that at low temperatures there may not be enough thermal energy to excite even one quantum of vibration. As a result, the oscillator stores very little energy, and the specific heat falls rapidly.
Einstein used this idea in his model of the specific heat of solids. Debye later improved the treatment by considering a spectrum of vibrational frequencies.
Hydrogen atom
The hydrogen atom was the central example of the old quantum theory. In the Bohr–Sommerfeld treatment, the angular part of the problem gives angular momentum quantum numbers, while the radial motion gives an additional quantum condition.
For a classical Kepler problem, the Hamiltonian can be written in terms of radial motion and angular momentum. Applying the old quantum condition gives energy levels depending mainly on the principal quantum number. This reproduced the Bohr energy formula and explained much of the hydrogen spectrum.
However, the model depended on classical orbits, which were later abandoned in modern quantum mechanics.
Space quantization and spin
The old quantum theory introduced the idea that angular momentum could be quantized along a chosen axis. This seemed paradoxical because it appeared to select a preferred direction in space.
The Stern–Gerlach experiment later demonstrated that atomic magnetic moments are quantized in a magnetic field. At first this seemed to confirm space quantization in the old theory, but the result was later understood in terms of electron spin, a feature not present in the original Bohr–Sommerfeld theory.
The discovery of spin and the Pauli exclusion principle helped explain the periodic table and marked the final stage of the old quantum theory.[14][15]
De Broglie waves
In 1924, Louis de Broglie proposed that matter has wave properties. He suggested that a particle with momentum has wavelength
or equivalently
This idea gave a physical interpretation to the old quantum condition: a classical orbit is allowed only when a matter wave fits around it as a standing wave. The phase change around the orbit must be an integer multiple of .
De Broglie's matter-wave hypothesis became one of the immediate bridges from the old quantum theory to modern wave mechanics.
BKS theory and the end of the old theory
In 1924, Bohr, Hendrik Kramers, and John C. Slater proposed the BKS theory, which treated atomic systems as quantum-like while keeping the electromagnetic field classical.[16]
The theory attempted to solve problems of radiation and atomic transitions without accepting strict energy conservation in individual microscopic events. It was soon rejected after the Bothe–Geiger coincidence experiment supported energy conservation in individual processes.[16]
At the same time, Kramers developed rules for calculating transition probabilities between quantum states. These ideas influenced Heisenberg's 1925 reformulation of quantum theory in terms of observable transition quantities, leading to matrix mechanics.
Transition to modern quantum mechanics
The old quantum theory ended when modern quantum mechanics was created in 1925–1926. Heisenberg reformulated quantum theory in terms of transition amplitudes between observable states, leading to matrix mechanics with Born and Jordan. Independently, Schrödinger developed wave mechanics, based partly on de Broglie's matter waves.
Schrödinger's equation reproduced the successes of the old quantum theory without relying on ambiguous classical orbits. Dirac later showed that matrix mechanics and wave mechanics were different forms of a more general transformation theory.
By the late 1920s, the old quantum theory had been replaced. Its successes were retained, but its classical-orbit picture was abandoned.
Limitations
The old quantum theory had several major limitations:[17]
- It could not calculate the intensities of spectral lines.
- It failed to explain the anomalous Zeeman effect without electron spin.
- It could not consistently describe multi-electron atoms.
- It worked only for separable or periodic classical systems.
- It could not quantize chaotic systems.
- It had no complete theory of measurement, probability, or wave functions.
Modern quantum mechanics explained these phenomena using operators, wave functions, Hilbert spaces, and probability amplitudes.
Legacy
Despite its limitations, the old quantum theory was historically essential. It introduced quantization, quantum numbers, atomic energy levels, and semi-classical reasoning. It also produced the first successful quantum models of radiation, specific heat, atomic spectra, and atomic structure.
The old theory is now understood as a semi-classical approximation to modern quantum mechanics. Related methods survive in the WKB approximation, the Einstein–Brillouin–Keller method, and semiclassical approaches to quantum chaos.[18][19][20]
See also
Table of contents (185 articles)
Index
Full contents
References
- ↑ Pais, Abraham (2005). Subtle is the Lord: The Science and the Life of Albert Einstein (illustrated ed.). OUP Oxford. p. 28. ISBN 978-0-19-280672-7. https://books.google.com/books?id=0QYTDAAAQBAJ.
- ↑ ter Haar, D. (1967). The Old Quantum Theory. Pergamon Press. pp. 206. ISBN 978-0-08-012101-7. https://archive.org/details/oldquantumtheory0000haar.
- ↑ "Semi-classical approximation". Encyclopedia of Mathematics. https://www.encyclopediaofmath.org/index.php?title=Semi-classical_approximation.
- ↑ Sakurai, J. J.; Napolitano, Jim (2014). "Quantum Dynamics". Modern Quantum Mechanics. Pearson. ISBN 978-1-292-02410-3.
- ↑ Kuhn, Thomas (1987). Black-Body Theory and the Quantum Discontinuity, 1894–1912. Chicago: University of Chicago Press. ISBN 0-226-45800-8.
- ↑
- Haas, Arthur Erich (1910). "Über die elektrodynamische Bedeutung des Planck'schen Strahlungsgesetzes und über eine neue Bestimmung des elektrischen Elementarquantums und der Dimension des Wasserstoffatoms" (in de). Sitzungsberichte der Kaiserlichen Akademie der Wissenschaften in Wien. Abt 2A 119: 119–144.
- Haas, A. E. (1909). Die Entwicklungsgeschichte des Satzes von der Erhaltung der Kraft (Habilitation Thesis) (in Deutsch). Vienna.
- Hermann, A. (1965) (in de). Arthur Erich Haas, Der erste Quantenansatz für das Atom. Stuttgart.
- ↑
- Nicholson, J. W. (1911). "The Spectrum of Nebulium". Monthly Notices of the Royal Astronomical Society 72: 49–64. doi:10.1093/mnras/72.1.49. Bibcode: 1911MNRAS..72...49N.
- Nicholson, J. W. (1911). "The Constitution of the Solar Corona. I.: Protofluorine". Monthly Notices of the Royal Astronomical Society 72 (2): 139–150. doi:10.1093/mnras/72.2.139. Bibcode: 1911MNRAS..72..139N.
- ↑ McCormmach, Russell (1966). "The Atomic Theory of John William Nicholson". Archive for History of Exact Sciences 3 (2): 160–184. doi:10.1007/BF00357268.
- ↑ Bohr, N. (1913). "On the constitution of atoms and molecules". The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science. Series 6 26 (151): 1–25. doi:10.1080/14786441308634955. Bibcode: 1913PMag...26....1B. https://zenodo.org/record/2493915.
- ↑ Sommerfeld, Arnold (1919). Atombau und Spektrallinien. Braunschweig: Friedrich Vieweg und Sohn.
- ↑ Pauling, Linus; Wilson, Edgar Bright (2012). Introduction to quantum mechanics: with applications to chemistry. New York: Dover Publications. ISBN 9780486134932. OCLC 830473042.
- ↑ Wilson, William (1915). "LXXXIII. The quantum-theory of radiation and line spectra". The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science 29 (174): 795–802. doi:10.1080/14786440608635362. https://zenodo.org/record/1430790.
- ↑ Sommerfeld, Arnold (1916). "Zur Quantentheorie der Spektrallinien" (in de). Annalen der Physik 356 (17): 1–94. doi:10.1002/andp.19163561702. Bibcode: 1916AnP...356....1S. https://zenodo.org/record/1424309.
- ↑ Kragh, Helge (1979). "Niels Bohr's Second Atomic Theory". Historical Studies in the Physical Sciences 10: 123–186. doi:10.2307/27757389.
- ↑ Kumar, Manjit (2008). Quantum: Einstein, Bohr, and the great debate about the nature of reality (1st American ed.). New York: W.W. Norton. Chap.7. ISBN 978-0-393-07829-9.
- ↑ 16.0 16.1 Bacciagaluppi, Guido; Valentini, Antony (2009-10-22). Quantum Theory at the Crossroads: Reconsidering the 1927 Solvay Conference. Cambridge University Press. ISBN 978-0-521-81421-8. https://www.google.fr/books/edition/Quantum_Theory_at_the_Crossroads/EAPX3JfQAgIC?hl=en&gbpv=1&dq=BKS+theory+bothe+old+quantum+theory&pg=PA86&printsec=frontcover.
- ↑ Chaddha, G.S. (2006). Quantum Mechanics. New Delhi: New Age International. pp. 8–9. ISBN 978-81-224-1465-3. https://books.google.com/books?id=Bzj2JcPeAHAC&pg=PA9.
- ↑ Stone, A.D. (August 2005). "Einstein's unknown insight and the problem of quantizing chaos". Physics Today 58 (8): 37–43. doi:10.1063/1.2062917. Bibcode: 2005PhT....58h..37S. https://www.eng.yale.edu/stonegroup/publications/phys_today.pdf.
- ↑ Solov'ev, E. A. (2011). "Classical approach in atomic physics". European Physical Journal D 65 (3): 331–351. doi:10.1140/epjd/e2011-20261-6. Bibcode: 2011EPJD...65..331S.
- ↑ Lev D. Landau; Evgeny M. Lifshitz (1977). Quantum Mechanics: Non-Relativistic Theory. 3 (3rd ed.). Pergamon Press. ISBN 978-0-08-020940-1.
Source attribution: Physics:Quantum mechanics/Timeline/Old quantum theory