Physics:Quantum Selection rules
Selection rules in quantum mechanics determine which transitions between quantum states are allowed or forbidden when an atom interacts with electromagnetic radiation. These rules arise from symmetry properties of the system and the structure of the interaction Hamiltonian.[1]
In atomic spectroscopy, the dominant mechanism is the electric dipole (E1) transition, which governs most observed spectral lines.
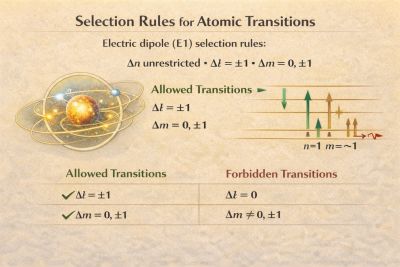
Electric dipole (E1) selection rules
For electric dipole transitions, the allowed changes in quantum numbers are:
- unrestricted
These rules follow from evaluating the dipole transition matrix elements between quantum states.[2]
---
Physical origin
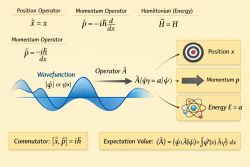
Selection rules arise from transition matrix elements of the form:
A transition is allowed only if this integral is non-zero. This condition is governed by symmetry principles such as parity and angular momentum conservation.[3]
---
Angular momentum considerations
The selection rules reflect conservation of angular momentum:
- A photon carries one unit of angular momentum
- Therefore:
The magnetic quantum number depends on photon polarization:
- (linear polarization)
- (circular polarization)
---
Forbidden transitions
Transitions that violate the E1 selection rules are called 'forbidden transitions. Typical examples include:
These transitions have vanishing electric dipole matrix elements and therefore very low probability.
However, they may occur via higher-order interactions:
- Magnetic dipole (M1) transitions
- Electric quadrupole (E2) transitions
These processes are much weaker but are important in astrophysical plasmas and precision spectroscopy.[4]
---
Spectroscopic consequences
Selection rules determine:
- Which spectral lines are observed
- The relative transition probabilities
- The polarization properties of emitted radiation
They are essential for interpreting atomic spectra and identifying elements in laboratory and astrophysical environments.[5]
---
Relation to hydrogen atom
In the hydrogen atom, selection rules explain the structure of spectral series such as:
- Lyman series ()
- Balmer series ()
Only transitions satisfying the E1 selection rules contribute significantly to observed spectra.
See also
Table of contents (185 articles)
Index
Full contents
References
Source attribution: Quantum Selection rules