Physics:Quantum Stationary states
A stationary state is a quantum state with all observables independent of time. It is an eigenvector of the energy operator (rather than a quantum superposition of different energies). It is also called an energy eigenstate, energy eigenfunction, or energy eigenket. Stationary states are fundamental in quantum mechanics and are closely related to concepts such as atomic orbitals and molecular orbitals.
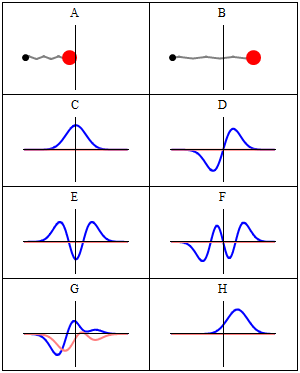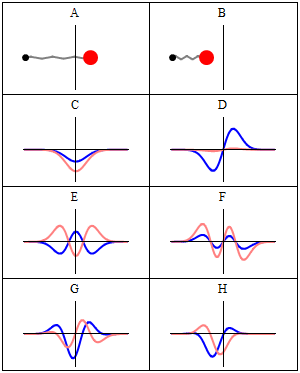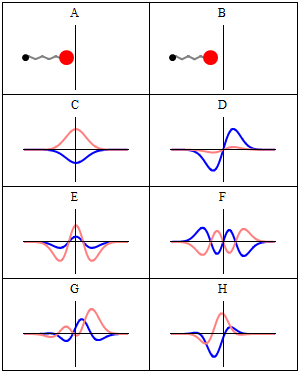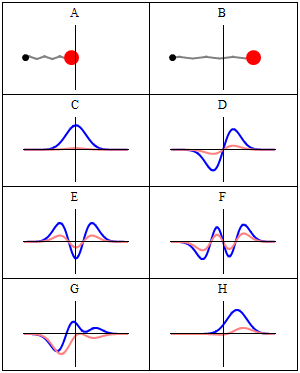
Introduction
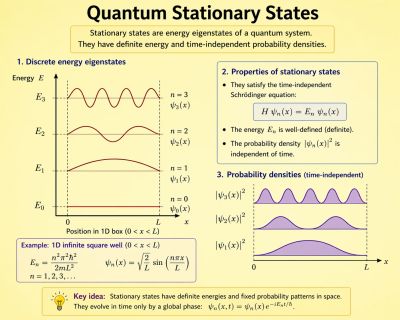
A stationary state is called stationary because the system remains unchanged in every observable way as time elapses. For a system with a time-independent Hamiltonian, this means that measurable quantities such as position probability, momentum, or spin remain constant in time.[1]
The wavefunction itself is not constant: it evolves by a global complex phase factor, forming a standing wave. The oscillation frequency of this phase, multiplied by the Planck constant, corresponds to the energy via the Planck–Einstein relation.
Definition
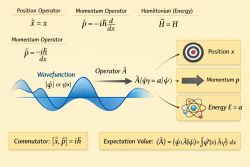
Stationary states are solutions to the time-independent Schrödinger equation:
where:
- is the quantum state,
- is the Hamiltonian operator,
- is the energy eigenvalue.
This is an eigenvalue equation: the stationary states are eigenvectors of the Hamiltonian.
When inserted into the time-dependent Schrödinger equation, the evolution is:[2]
with solution:
Thus, a stationary state evolves only by a phase factor, with angular frequency .
Stationary state properties
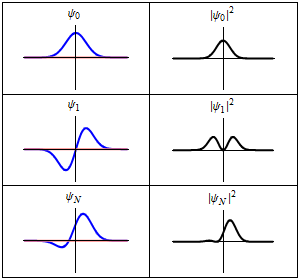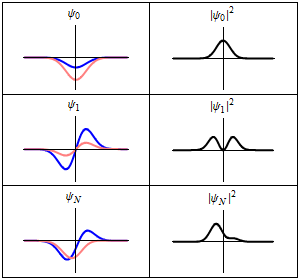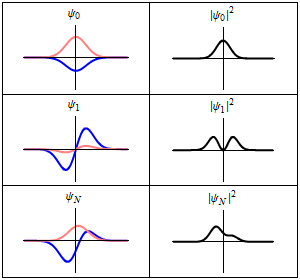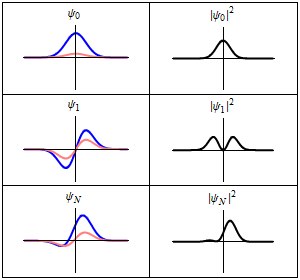
Although the wavefunction changes in time,
all observable quantities remain constant. For example, the probability density:
is time-independent.
In the Heisenberg picture, stationary states are mathematically constant in time.
These results assume a time-independent Hamiltonian; if the system changes, the state will generally no longer be stationary.
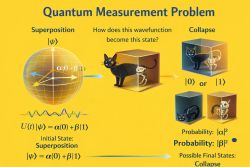
Superposition
A general quantum state need not be stationary. A superposition of stationary states with different energies leads to time-dependent interference effects, producing a changing probability distribution.
Spontaneous decay
In ideal (nonrelativistic) quantum mechanics, systems such as the hydrogen atom have many stationary states. However, in reality, excited states are not perfectly stationary: an electron in a higher energy level can undergo spontaneous emission, emitting a photon and decaying to a lower-energy state.[3]
This occurs because the usual Hamiltonian is an approximation; more complete descriptions from quantum field theory include effects such as vacuum fluctuations, which break exact stationarity for excited states.
Comparison to orbitals
An atomic orbital or molecular orbital can be interpreted as a stationary state (or approximation thereof) for a single electron.[4]
For single-electron systems (such as hydrogen), orbitals correspond directly to stationary states. For many-electron systems, however, the full stationary state is a many-particle state, often approximated using methods such as Slater determinants.[5]
In practice, orbitals are useful approximations based on treating electrons independently (the single-electron approximation), often combined with the Born–Oppenheimer approximation.
See also
Table of contents (185 articles)
Index
Full contents
References
- ↑ Claude Cohen-Tannoudji, Bernard Diu, and Franck Laloë. Quantum Mechanics: Volume One. Hermann, 1977. p. 32.
- ↑ Quanta: A handbook of concepts, P. W. Atkins, Oxford University Press, 1974, Template:ISBN.
- ↑ Quantum Physics of Atoms, Molecules, Solids, Nuclei, and Particles (2nd Edition), R. Eisberg, R. Resnick, John Wiley & Sons, 1985, Template:ISBN
- ↑ Physical chemistry, P. W. Atkins, Oxford University Press, 1978, Template:ISBN.
- ↑ Löwdin, Per-Olov (1955). "Quantum theory of many-particle systems". Physical Review 97 (6): 1474–1489.
Source attribution: Physics:Stationary state