Physics:Quantum Wave–particle duality
Wave–particle duality is the concept in quantum mechanics that quantum entities exhibit particle-like or wave-like properties depending on the experimental circumstances.[1][2] It expresses the failure of purely classical categories such as ‘‘particle’’ and ‘‘wave’’ to give a complete description of quantum objects.[1][2]
During the 19th and early 20th centuries, light was first understood as a wave and later shown to possess a particulate character, while electrons were initially treated as particles and later found to exhibit diffraction and interference. The term wave–particle duality arose to describe these apparently contradictory behaviors.
History
In the late 17th century, Isaac Newton argued that light consisted of particles, whereas Christiaan Huygens proposed a wave theory of light.[4] In the early 19th century, Thomas Young's interference experiments and François Arago's observation of the Poisson spot strongly supported the wave picture of light.
That picture was challenged by Planck's law for black-body radiation, which required energy exchange in discrete quanta.[5] In 1905, Albert Einstein interpreted the photoelectric effect using discrete light quanta, later called photons.[6] The case for particle-like light was greatly strengthened by Arthur Compton's measurements of photon momentum in scattering experiments.[7]
For electrons, the historical sequence was reversed. Their charge-to-mass ratio and trajectories in electromagnetic fields supported a particle picture.[8] In 1924, Louis de Broglie proposed that matter has an associated wavelength, relating a particle’s momentum to a wave description.[9] This idea rapidly led to wave mechanics and the Schrödinger equation.[10]
Experimental confirmation soon followed in the Davisson–Germer experiment and in the diffraction experiments of George Paget Thomson and Alexander Reid, showing that electrons produce diffraction patterns characteristic of waves.[11][12]
Classical waves and particles
In classical physics, waves and particles are distinct models with different mathematical descriptions. Classical waves obey wave equations, extend through space, diffract, and interfere. Examples include water waves, sound waves, and radio waves. Classical particles follow trajectories specified by position and velocity, and in the absence of forces they move in straight lines. Planets, sand grains, and bullets are well described by particle models.
Quantum systems do not fit neatly into either classical category. The wavefunction evolves according to a wave equation, but measurements yield discrete detection events associated with particles. The probability of finding a quantum object at a given location is determined by the squared magnitude of a complex probability amplitude.[1] Thus many repeated particle detections can build up an interference pattern that reflects wave-like evolution.
Electrons behaving as waves and particles
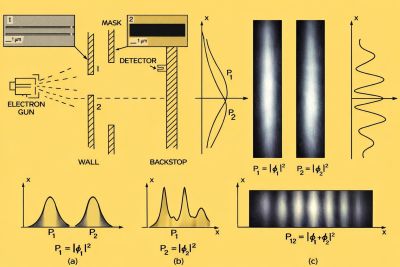
The electron double-slit experiment is one of the clearest demonstrations of wave–particle duality.[2][3] Electrons emitted toward two narrow slits form a diffraction pattern when one slit is open and an interference pattern when both slits are open. At high intensity the detector records a smooth pattern, but when the beam intensity is reduced so that electrons arrive one at a time, each detection appears as an individual localized dot. Over time, those dots accumulate into the same interference pattern predicted by wave mechanics.[3]
This combination of localized impacts and wave-like interference is not limited to electrons. Comparable interference has been demonstrated for atoms and large molecules, showing that wave–particle duality is a general feature of quantum matter.[13]
Observing photons as particles
For photons, the historical path ran in the opposite direction. Light was long understood as a wave, but the photoelectric effect showed that energy transfer from light to matter occurs in discrete units.[14][15] Einstein proposed that light of frequency is absorbed or emitted in quanta of energy
where is the Planck constant.[6] This relation explains why electrons are emitted only when the photon frequency exceeds a threshold set by the material’s work function. Increasing the light intensity below that threshold does not liberate electrons, whereas increasing the photon frequency above threshold increases the maximum kinetic energy of emitted electrons.
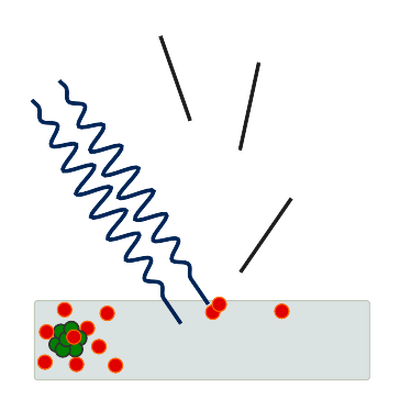
Further evidence for photon-like behavior came from Compton scattering, where X-rays exchange momentum with electrons in a way consistent with particle collisions.[7] Thus light exhibits interference and diffraction characteristic of waves, while also carrying discrete energy and momentum characteristic of particles.
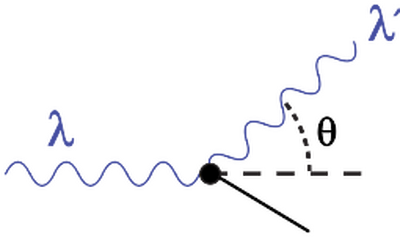
Duality with other properties
Wave–particle duality is part of a broader quantum pattern in which classical attributes do not remain cleanly separated. Angular momentum, for example, is often pictured as a particle-like quantity, but electromagnetic waves can carry orbital angular momentum when arranged in vortex beams.[16] Likewise, electron waves can be prepared with large orbital angular momentum and still display diffraction, interference, and magnetic interactions associated with particle properties.[17][18]
File:Focused Laguerre-Gaussian beam.webm
Which-slit experiments
In “which-slit” or “which-way” experiments, detectors are placed so that the path taken by a quantum particle can be inferred. Quantum mechanics predicts that when path information becomes available, the interference pattern disappears because coherence between the alternatives is lost.[2] Many realizations of this idea, including delayed-choice experiments, confirm the same principle: path information and interference visibility are mutually constrained.[19]
A standard optical realization uses a Mach–Zehnder interferometer. A beam is split into two paths and later recombined. When the second beam splitter is present, the outputs depend on interference between the two paths. When it is removed, each output reveals path information and the interference effect vanishes.[20]
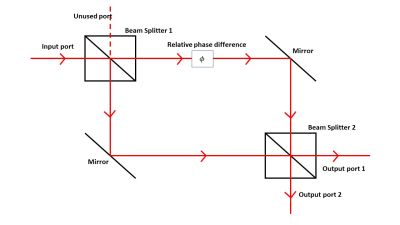
Significance
Wave–particle duality does not mean that quantum objects alternate between being literal classical waves and literal classical particles. Rather, quantum theory assigns them a state described by a wavefunction or probability amplitude, while measurements yield discrete outcomes associated with quanta. The apparent duality reflects the limits of classical language and the need for a distinctly quantum description.[1][2]
See also
Table of contents (185 articles)
Index
Full contents
References
- ↑ 1.0 1.1 1.2 1.3 Messiah, Albert (1966). Quantum Mechanics. North Holland, John Wiley & Sons. ISBN 0486409244. https://archive.org/details/quantummechanics0000mess/quantummechanics0000mess.
- ↑ 2.0 2.1 2.2 2.3 2.4 Feynman, Richard P.; Leighton, Robert B.; Sands, Matthew L. (2007). The Feynman Lectures on Physics, Volume III: Quantum Mechanics. Addison-Wesley. ISBN 978-0-201-02118-9. https://www.feynmanlectures.caltech.edu/III_toc.html.
- ↑ 3.0 3.1 3.2 Bach, Roger; Pope, Damian; Liou, Sy-Hwang; Batelaan, Herman (2013-03-13). "Controlled double-slit electron diffraction". New Journal of Physics (IOP Publishing) 15 (3): 033018. doi:10.1088/1367-2630/15/3/033018. ISSN 1367-2630. Bibcode: 2013NJPh...15c3018B. https://iopscience.iop.org/article/10.1088/1367-2630/15/3/033018.
- ↑ Huygens, Christiaan (1690). Traité de la lumiere. Leiden: Pieter van der Aa.
- ↑ Planck, Max (1901). "Ueber das Gesetz der Energieverteilung im Normalspectrum" (in de). Annalen der Physik 309 (3): 553–563. doi:10.1002/andp.19013090310.
- ↑ 6.0 6.1 Einstein, Albert (1905). "Über einen die Erzeugung und Verwandlung des Lichtes betreffenden heuristischen Gesichtspunkt". Annalen der Physik 17 (6): 132–148. doi:10.1002/andp.19053220607.
- ↑ 7.0 7.1 Whittaker, Edmund T. (1989). A history of the theories of aether & electricity. 2: The modern theories, 1900–1926. New York: Dover Publications. ISBN 978-0-486-26126-3.
- ↑ Thomson, J. J. (1897). "XL. Cathode Rays". The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science 44 (269): 293–316. doi:10.1080/14786449708621070.
- ↑ de Broglie, Louis Victor. "On the Theory of Quanta". https://fondationlouisdebroglie.org/LDB-oeuvres/De_Broglie_Kracklauer.pdf.
- ↑ Schrödinger, E. (1926). "An Undulatory Theory of the Mechanics of Atoms and Molecules". Physical Review 28 (6): 1049–1070. doi:10.1103/PhysRev.28.1049. Bibcode: 1926PhRv...28.1049S.
- ↑ Davisson, C.; Germer, L. H. (1927). "Diffraction of Electrons by a Crystal of Nickel". Physical Review 30 (6): 705–740. doi:10.1103/PhysRev.30.705.
- ↑ Thomson, G. P.; Reid, A. (1927). "Diffraction of Cathode Rays by a Thin Film". Nature 119 (3007): 890. doi:10.1038/119890a0.
- ↑ Arndt, Markus; Hornberger, Klaus (2014). "Testing the limits of quantum mechanical superpositions". Nature Physics 10 (4): 271–277. doi:10.1038/nphys2863.
- ↑ Whittaker, E. T. (1910). A History of the Theories of Aether and Electricity: From the Age of Descartes to the Close of the Nineteenth Century. Longman, Green and Co..
- ↑ Wheaton, Bruce R. (1978). "Philipp Lenard and the Photoelectric Effect, 1889-1911". Historical Studies in the Physical Sciences 9: 299–322. doi:10.2307/27757381.
- ↑ Allen, L.; Beijersbergen, M. W.; Spreeuw, R. J. C.; Woerdman, J. P. (1992). "Orbital angular momentum of light and the transformation of Laguerre-Gaussian laser modes". Physical Review A 45 (11): 8185–8189. doi:10.1103/PhysRevA.45.8185. PMID 9906912.
- ↑ Verbeeck, J.; Tian, H.; Schattschneider, P. (2010). "Production and application of electron vortex beams". Nature 467 (7313): 301–304. doi:10.1038/nature09366. PMID 20844532.
- ↑ Tavabi, A. H.; Rosi, P.; Roncaglia, A.; Rotunno, E.; Beleggia, M.; Lu, P.-H.; Belsito, L.; Pozzi, G. et al. (2022). "Generation of electron vortex beams with over 1000 orbital angular momentum quanta using a tunable electrostatic spiral phase plate". Applied Physics Letters 121 (7). doi:10.1063/5.0093411.
- ↑ Ma, Xiao-song; Kofler, Johannes; Zeilinger, Anton (2016). "Delayed-choice gedanken experiments and their realizations". Reviews of Modern Physics 88 (1): 015005. doi:10.1103/RevModPhys.88.015005.
- ↑ Schneider, Mark B.; LaPuma, Indhira A. (2002). "A simple experiment for discussion of quantum interference and which-way measurement". American Journal of Physics 70 (3): 266–271. doi:10.1119/1.1450558. https://digital.grinnell.edu/islandora/object/grinnell%3A47/datastream/OBJ/download/A_Simple_Experiment_for_Discussion_of_Quantum_Interference_and_Which-Way_Measurement.pdf.
Source attribution: Physics:Quantum Wave–particle duality