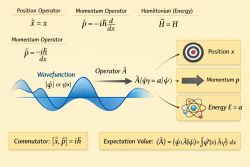
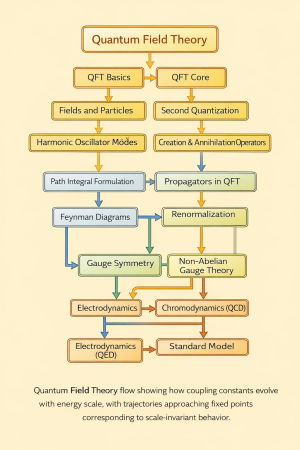
Physics:Quantum Distribution functions
Quantum distribution functions describe the average occupation of energy states in a many-particle system at thermal equilibrium. They distinguish classical from quantum statistical behavior.
For a state of energy , the occupation depends on particle type.[1]
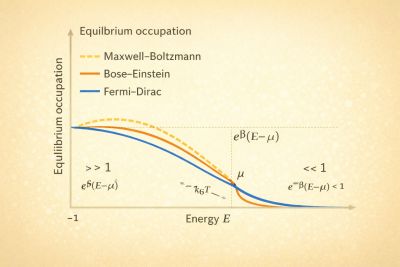
Maxwell–Boltzmann distribution
In the classical limit:
Valid when quantum degeneracy is negligible.[2]
Bose–Einstein distribution
For bosons:
Bosons can accumulate in low-energy states, leading to Bose–Einstein condensation.[3]
Fermi–Dirac distribution
For fermions:
The Pauli exclusion principle limits occupation to one particle per state.[4]
At low temperature, the distribution approaches a step function at the Fermi energy.
Classical limit
When , both quantum distributions reduce to:
Chemical potential
The chemical potential controls particle number.
- For fermions: at low temperature
- For bosons:
These constraints determine quantum gas behavior.[1]
Physical interpretation
The three distributions reflect different statistics:
- Maxwell–Boltzmann → classical limit
- Bose–Einstein → state clustering
- Fermi–Dirac → exclusion principle
These differences produce distinct macroscopic phenomena.[1]
Applications
Quantum distribution functions are essential in:
- classical gases and kinetic theory[2]
- electron behavior in solids[4]
- photons and phonons[3]
- quantum many-body systems[1]
See also
Table of contents (185 articles)
Index
Full contents
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 https://www.damtp.cam.ac.uk/user/tong/statphys/statmechhtml/S3.html
- ↑ 2.0 2.1 2.2 2.3 https://ocw.mit.edu/courses/5-62-physical-chemistry-ii-spring-2008/2351f20e4727ae0a7e03ccaca02452d7_08_562ln08.pdf
- ↑ 3.0 3.1 https://ocw.mit.edu/courses/8-08-statistical-physics-ii-spring-2005/resources/the_bose_gas/
- ↑ 4.0 4.1 https://ocw.mit.edu/courses/8-08-statistical-physics-ii-spring-2005/3d0cf2cb43a2b62f92089db14e8e2904_the_fermi_gas.pdf
Source attribution: Quantum Distribution functions