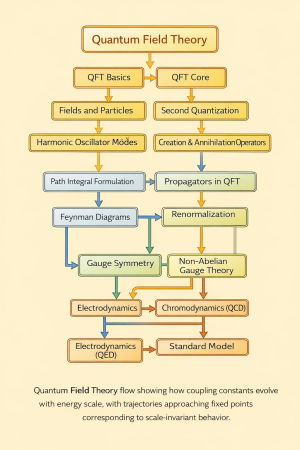
Physics:Quantum Multi-electron atoms
Multi-electron atoms are atomic systems containing more than one electron, where the simple analytical solutions of the hydrogen atom no longer apply. The presence of multiple electrons introduces Coulomb interactions between electrons, leading to complex energy structures and requiring approximation methods to solve the Schrödinger equation.[1]
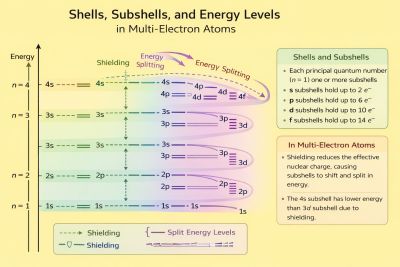
Electron–electron interactions
In multi-electron atoms, each electron interacts not only with the nucleus but also with all other electrons. The Hamiltonian takes the form:
where the second summation represents electron–electron repulsion.[2]
This interaction prevents exact analytical solutions and leads to correlated electron motion.
Screening and effective nuclear charge
Electrons partially shield each other from the nuclear charge. As a result, each electron experiences an effective nuclear charge , which is smaller than the actual nuclear charge . This effect explains:
- Orbital energy ordering
- Periodic trends in atomic structure
- Variations in ionization energies
Screening leads to deviations from hydrogen-like energy levels.[3]
Approximation methods
Because exact solutions are not possible, several approximation techniques are used:
Hartree and Hartree–Fock methods
These methods approximate the total wavefunction as a product (or antisymmetrized product) of single-electron orbitals. The Hartree–Fock method includes exchange effects arising from electron indistinguishability.[4]
Central field approximation
The atom is approximated as electrons moving in an average spherically symmetric potential, simplifying the many-body problem.
Configuration interaction
Improves accuracy by combining multiple electron configurations to account for electron correlation effects.
Pauli principle and exchange symmetry
Electrons are fermions and must obey the Pauli exclusion principle. The total wavefunction must be antisymmetric under particle exchange:
This requirement leads to:
- Electron shell structure
- Spin pairing
- Exchange energy contributions
Spectral structure and complexity
Multi-electron atoms exhibit:
- Fine and hyperfine structure splitting
- Complex spectral line patterns
- Term symbols describing total angular momentum
These features arise from combined effects of electron interactions, spin–orbit coupling, and external perturbations.[5]
Significance
Understanding multi-electron atoms is essential for:
- Atomic spectroscopy
- Chemistry and bonding
- Laser physics
- Astrophysical spectra interpretation
They form the foundation for real atomic systems beyond hydrogen and connect quantum mechanics to observable material properties.
See also
Table of contents (185 articles)
Index
Full contents
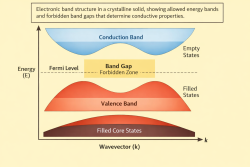
References
- ↑ Griffiths, D. J. (2018). Introduction to Quantum Mechanics. Cambridge University Press.
- ↑ Sakurai, J. J. (2017). Modern Quantum Mechanics. Cambridge University Press.
- ↑ Atkins, P. (2018). Physical Chemistry. Oxford University Press.
- ↑ Szabo, A.; Ostlund, N. S. (1996). Modern Quantum Chemistry. Dover.
- ↑ Bransden, B. H.; Joachain, C. J. (2003). Physics of Atoms and Molecules. Pearson.
Source attribution: Physics:Quantum Multi-electron atoms