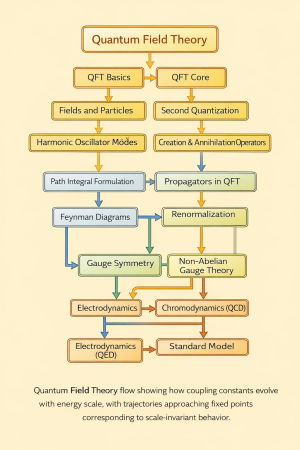
Physics:Quantum Boundary conditions and quantization
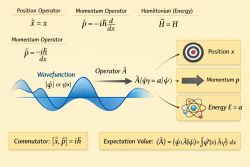
Quantum boundary conditions and quantization describe how physical constraints on wavefunctions restrict the allowed solutions of the Schrödinger equation, leading to discrete energy levels.[1]
Boundary conditions
Wavefunctions must satisfy specific physical conditions:
- Continuity of
- Finite values everywhere
- Boundary values imposed by the physical system
- Vanishing at infinite potential walls
These conditions ensure physically meaningful probability distributions.[2]
Quantization from confinement
A fundamental example is a particle confined in a one-dimensional box of length :
- Boundary conditions: ,
- Allowed solutions:
Only discrete values of satisfy these conditions.
This leads directly to quantized energy levels.[3]
Energy quantization
The allowed energies for a particle in a box are:
where:
- is a positive integer
- is the particle mass
- is the size of the system
Energy becomes discrete because only standing-wave solutions compatible with the boundaries are allowed.[4]
Physical interpretation
Quantization arises because:
- Only wavefunctions that “fit” within the boundaries are allowed
- Standing-wave solutions form discrete modes
- Continuous classical motion is replaced by discrete allowed states
This explains why confined quantum systems exhibit discrete spectra.[5]
Generalization
Boundary-condition-induced quantization occurs in many systems:
- Atoms (electron orbitals)
- Molecules (vibrational modes)
- Quantum wells and nanostructures
- Electromagnetic cavity modes
In each case, constraints produce discrete spectra.[6]
Applications
Quantization due to boundary conditions is central to:
- Atomic spectra
- Semiconductor devices
- Nanotechnology
- Quantum confinement effects
Allowed energy levels and transitions underlie spectroscopy and quantum devices.[7]
See also
Table of contents (185 articles)
Index
Full contents
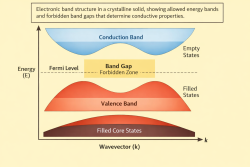
References
Source attribution: Quantum Boundary conditions and quantization