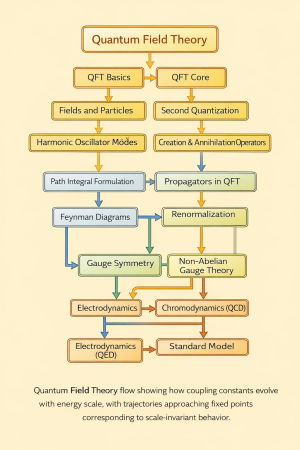
Physics:Quantum Spectral lines and series
Spectral lines are discrete wavelengths of light emitted or absorbed by atoms and molecules, arising from transitions between quantized energy levels, and they provided some of the earliest direct evidence for quantum theory.[1]

Origin of spectral lines
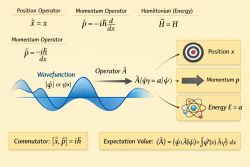
In quantum mechanics, electrons in atoms occupy discrete energy eigenstates. When an electron transitions between two states, a photon is emitted or absorbed with energy given by:
where:
- is Planck’s constant
- is the frequency of the radiation
- , are the initial and final energy levels
This leads to sharply defined spectral lines rather than a continuous spectrum.[2]
Hydrogen spectral series
The hydrogen atom provides the simplest and most important example of spectral line structure. Its energy levels are given by:
Transitions between these levels produce series of spectral lines described by the Rydberg formula:
where:
- is the Rydberg constant
This relation accurately predicts observed hydrogen spectral lines.[3]
Major series
- Lyman series () – ultraviolet region
- Balmer series () – visible region
- Paschen series () – infrared region
- Brackett series () – infrared
- Pfund series () – far infrared
Each series corresponds to transitions ending at a fixed lower energy level.[4]
Fine structure and splitting
Real spectral lines are not perfectly sharp. They exhibit splitting due to additional physical effects:
- Fine structure — relativistic corrections and spin–orbit coupling
- Zeeman effect — splitting in an external magnetic field
- Stark effect — splitting in an electric field
These effects reveal deeper structure in atomic energy levels.[5][6]
Selection rules
Not all transitions are allowed. Selection rules determine which spectral lines appear:
These arise from conservation of angular momentum and symmetry properties of atomic wavefunctions.[7]
Spectroscopy and applications
Spectral lines are fundamental in many areas of physics and astronomy:
- Identifying chemical elements in stars and galaxies
- Measuring Doppler shifts and cosmic expansion
- Determining temperatures and densities of plasmas
- Laser technology and atomic clocks
Each element has a unique spectral “fingerprint.”[8]
See also
Table of contents (185 articles)
Index
Full contents
References
Source attribution: Quantum Spectral lines and series