Physics:Quantum The Secret of Cohesion and How Waves Hold Matter Together
Overview: Quantum Mechanisms of Cohesion
Classical vs Quantum Explanation for Matter Stability

Opposite charges attract, but that explanation cannot account for the stability of atoms.
If electrons were point particles, they would collapse into the nucleus.
The stability of matter is quantum mechanical.
Electrons are described by wavefunctions.
These waves:
- Spread through space
- Interfere with one another
- Obey symmetry principles such as the Pauli exclusion principle
Waves do not hold matter together by acting like glue. Instead, cohesion arises from the overlap, interference, and symmetry constraints of quantum wavefunctions, together with the balance between kinetic energy and Coulomb forces.
In this course we examine how:
- The Schrödinger equation already encodes stability
- Wavefunction overlap produces chemical bonds
- Fermi statistics prevents collapse
- Collective coherence gives rise to metals and superconductors
- Decoherence in open systems can modify binding
Matter holds together because particles behave as quantum waves.
Classical vs Quantum Picture.
- Cohesion in matter exists because particles have wave character, and wavefunctions overlap, interfere, and obey symmetry constraints.
Let’s build this step by step.
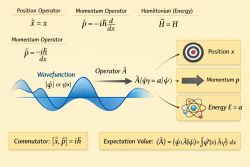
1️⃣ Starting Point: The Schrödinger Equation

The Schrödinger equation a partial differential equation that overlooks the wave function of non-relativistic quantum-mechanical systems.[1]Template:Rp Its discovery was significant in the development of quantum mechanics. It is named after Erwin Schrödinger, who postulated the equation in 1925 and published it in 1926. It forms the basis for the work that leaded to his Nobel Prize in Physics in 1933.[2][3][4]
For an electron in a potential :
The first term,
represents the kinetic energy operator and is purely wave-like (it depends on the spatial curvature of the wavefunction).
The second term,
represents the interaction energy (e.g., the Coulomb potential).
Cohesion emerges from the interplay between:
- Wave-like delocalization (kinetic term )
- Coulomb attraction between charges encoded in

2️⃣ Wave Nature of Particles and Quantum Stability

Matter waves, a central part of the theory of quantum mechanics, being half of wave–particle duality. All scales where measurements have been practical, matter exhibits wave-like behavior. A beam of electrons can be diffracted like a beam of light or a water wave.
Matter behaves like a wave was proposed by French physicist Louis de Broglie in 1924, and matter waves are also known as de Broglie waves.
If electrons were classical point particles, they would collapse into the nucleus and atoms would not be stable.
To find the wavelength equivalent to a moving body, de Broglie set the total energy from special relativity for that body equal to Template:Math:
(Modern physics no longer uses this form of the total energy; the Wikipedia:energy–momentum relation:energy–momentum relation has proven more useful.) De Broglie identified the velocity of the particle, , with the wave group velocity in free space:
(The modern definition of group velocity uses angular frequency Template:Mvar and wave number Template:Mvar). By applying the differentials to the energy equation and identifying the relativistic momentum:
then integrating, de Broglie arrived at his formula for the relationship between the wavelength, Template:Mvar, associated with an electron and the modulus of its momentum, Template:Math, through the Planck constant, Template:Math:[5] The quantum kinetic energy associated with spatial confinement scales as
where is the characteristic localization length.
As becomes smaller (stronger localization), the kinetic energy increases rapidly. This follows from the Laplacian term in the Schrödinger equation and is closely related to the uncertainty principle,
Thus, wave behavior prevents atomic collapse.
3️⃣ Covalent Bonding and Wavefunction Overlap
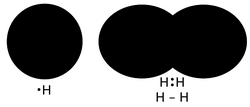
Covalent bonding a chemical bond that involves electrons to form electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs. Stable balance of attracting and repulsive forces between atoms, when they share electrons, is known as covalent bonding.[6] For molecules, to share electrons allows each atom to attain the equivalent of a full valence shell, responding to an electronic configuration. In organic chemistry, covalent bonding is more common than ionic bonding.
Consider two hydrogen atoms. Their atomic orbitals can combine into symmetric and antisymmetric superpositions:
Bonding combination:
Antibonding combination:
The bonding state has increased probability density between the nuclei:
in the internuclear region.
This lowers the total energy through:
- Electron delocalization
- Reduced Coulomb repulsion
- Lower effective kinetic energy due to spreading
Bonding is therefore an energetic consequence of constructive wave interference.
4️⃣ Fermi Statistics and Pauli Effects
A degenerate Fermi gas represents a state of matter where particles (fermions) are packed at high density and low temperatures, causing quantum effects. The pressure of a degenerate Fermi gas, known as degeneracy pressure, differs between high-density (high-pressure) and low-density (low-pressure) regimes, because the pressure depends on density () rather than temperature ()
Electrons are fermions, so the total wavefunction must be antisymmetric under exchange:
This implies the Pauli exclusion principle:
for each single-particle quantum state .
As electrons fill higher momentum states, the Fermi momentum increases, giving rise to Fermi pressure. The associated energy density scales as
This quantum pressure stabilizes matter and prevents collapse.
5️⃣ Exchange Interaction and Collective Order

Antisymmetry, the total energy includes an exchange term. In simplified form, the exchange contribution between two states can be written as
This term has no classical analogue and arises purely from wavefunction symmetry.
It explains phenomena such as:
- Ferromagnetism
- Antiferromagnetism
- Additional molecular stabilization
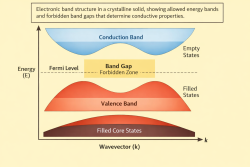
6️⃣ Metallic Bonding: Bloch Waves
Metallic bonding is a chemical bonding that arises from the electrostatic attractive force between conduction electrons (in the form of an electron cloud of delocalized electrons) and positively charged metal ions. It is the sharing of free electrons among a crystal structure of positively charged ions (cations). Metallic bonding accounts for physical properties of metals, such as strength, ductility, thermal and Wikipedia:electrical resistivity and conductivity, opacity, and lustre.[7][8][9][10]
In a periodic lattice potential , electron eigenstates take the Bloch form:
where
for lattice vectors .
These delocalized wave functions extend across the crystal and lower total energy collectively, producing metallic cohesion.
7️⃣ Superconductivity as a Cohesive Quantum Phenomenon
Superconductivity, physical properties observed in superconductors: materials where electrical resistance vanishes and magnetic fields are expelled from the material. Unlike a standard metallic conductor, whose resistance decreases gradually as its temperature is lowered, even down to near absolute zero, a superconductor characteristics are critical temperature below which the resistance drops abruptly to zero.[11][12] An electric current through a loop of superconducting wire can persist indefinitely with no power source.[13][14][15][16]
Through electron-phonon interaction, an effective attractive interaction can arise:
This leads to Cooper pairing and a macroscopic coherent state with order parameter
Cohesion here emerges from collective quantum phase coherence.
8️⃣ Stability of Matter: Mathematical Perspective
Stability of matter refers to the ability of a large number of charged particles, such as electrons and protons, to create macroscopic objects without collapsing or blowing apart due to electromagnetic interactions. Classical physics predicts that such systems should be inherently unstable due to attractive and repulsive electrostatic forces between charges, and thus the stability of matter was a theoretical problem that required a quantum mechanical explanation.
The first solution to this problem was provided by Freeman Dyson and Andrew Lenard in Freeman Dyson 1967–1968,[17][18] but a shorter and more conceptual proof was found later by Elliott Lieb in 1975 using the Lieb–Thirring inequality.[19] The stability of matter is partly due to the uncertainty principle and the Pauli exclusion principle.[20]
The stability of matter results from the balance:
The kinetic term originates from wave curvature and the Coulomb term from electromagnetic interaction.
Without the quantum kinetic term, the energy would decrease without bound and matter would collapse.
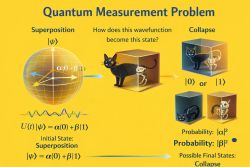
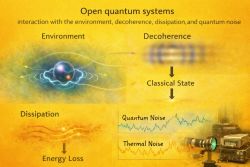
9️⃣ Decoherence Effects on Binding in Open Quantum Systems.

In physics, an open quantum system is a quantum mechanical system that interacts with an external quantum system, "the environment" or a "bath". In general, interactions significantly change the dynamics of the system, the information contained in the system is then lost to its environment. No quantum system is completely isolated from its surroundings,[21] it is important to develop a theoretical framework for treating these interactions to obtain an accurate understanding of quantum systems.
In open systems:
- Cohesion is related to coherence.
- Redfield/Lindblad dynamics describe loss of coherence.
- Decoherence can influence binding (e.g., exciton dynamics).
Under strong dissipation:
- Delocalization decreases
- Effective binding can change
So:
- Cohesion is sensitive to quantum coherence.
🔬 Summary: What Holds Matter Together?
Do waves in matter create cohesion?
Not as classical vibrations.
But at the quantum level:
- Yes — the wave character of electrons is essential for binding.
- Interference, antisymmetry, and delocalization make stable matter possible.
- Without quantum wave mechanics, matter would not exist.
See also
Table of contents (185 articles)
Index
Full contents
References
- ↑ Griffiths, David J. (2004). Introduction to Quantum Mechanics (2nd ed.). Prentice Hall. ISBN 978-0-13-111892-8.
- ↑ Template:Cite news
- ↑ Schrödinger, E. (1926). "An Undulatory Theory of the Mechanics of Atoms and Molecules". Physical Review 28 (6): 1049–70. doi:10.1103/PhysRev.28.1049. Bibcode: 1926PhRv...28.1049S. http://home.tiscali.nl/physis/HistoricPaper/Schroedinger/Schroedinger1926c.pdf.
- ↑ E Schrödinger, Quantisierung als Eigenwertproblem, "Erste Mitteilung", 'Ann Phys' 79 (1926) 361
- ↑ McEvoy, J. P.; Zarate, Oscar (2004). Introducing Quantum Theory. Totem Books. pp. 110–114. ISBN 978-1-84046-577-8.
- ↑ Whitten, Kenneth W.; Gailey, Kenneth D.; Davis, Raymond E. (1992). "7-3 Formation of covalent bonds". General Chemistry (4th ed.). Saunders College Publishing. p. 264. ISBN 0-03-072373-6.
- ↑ Metallic bonding. chemguide.co.uk
- ↑ Metal structures. chemguide.co.uk
- ↑ Chemical Bonds. chemguide.co.uk
- ↑ "Physics 133 Lecture Notes" Spring, 2004. Marion Campus. physics.ohio-state.edu
- ↑ Combescot, Roland (2022). Superconductivity. Cambridge University Press. pp. 1–2. ISBN 978-1-108-42841-5. https://books.google.com/books?id=lRhdEAAAQBAJ&pg=PA1.
- ↑ Fossheim, Kristian; Sudboe, Asle (2005). Superconductivity: Physics and Applications. John Wiley and Sons. pp. 7. ISBN 978-0-470-02643-4. https://books.google.com/books?id=Ep1MLS9YQX8C&pg=PA13.
- ↑ Bardeen, John; Cooper, Leon; Schrieffer, J. R. (December 1, 1957). "Theory of Superconductivity". Physical Review 108 (5): 1175. doi:10.1103/physrev.108.1175. Bibcode: 1957PhRv..108.1175B. https://books.google.com/books?id=_QKPGDG-cuAC&q=%22persist+indefinitely&pg=PA76. Retrieved June 6, 2014. Reprinted in Bogoliubov, Nikolaĭ Nikolaevich, ed (1963). The Theory of Superconductivity. 4. Gordon and Breach. p. 73. OCLC 537010. https://books.google.com/books?id=_QKPGDG-cuAC&pg=PA73.
- ↑ Daintith, John (2009) (in en). The Facts on File Dictionary of Physics (4th ed.). Infobase Publishing. p. 238. ISBN 978-1-4381-0949-7. https://books.google.com/books?id=VdEVdJo3CDgC&pg=PA238.
- ↑ Gallop, John C. (1990) (in en). SQUIDS, the Josephson Effects and Superconducting Electronics. CRC Press. pp. 1, 20. ISBN 978-0-7503-0051-3. https://books.google.com/books?id=ad8_JsfCdKQC.
- ↑ Durrant, Alan (2000). Quantum Physics of Matter. CRC Press. pp. 102–103. ISBN 978-0-7503-0721-5. https://books.google.com/books?id=F0JmHRkJHiUC&q=%22persist+indefinitely&pg=PA103.
- ↑ Dyson, Freeman J.; Lenard, A. (March 1967). "Stability of Matter. I". Journal of Mathematical Physics 8 (3): 423–434. doi:10.1063/1.1705209. Bibcode: 1967JMP.....8..423D.
- ↑ Lenard, A.; Dyson, Freeman J. (May 1968). "Stability of Matter. II". Journal of Mathematical Physics 9 (5): 698–711. doi:10.1063/1.1664631. Bibcode: 1968JMP.....9..698L.
- ↑ Lieb, Elliott H.; Thirring, Walter E. (15 September 1975). "Bound for the Kinetic Energy of Fermions Which Proves the Stability of Matter". Physical Review Letters 35 (11): 687–689. doi:10.1103/PhysRevLett.35.687. Bibcode: 1975PhRvL..35..687L.
- ↑ Marder, Michael P. (2010-11-17) (in en). Condensed Matter Physics. John Wiley & Sons. ISBN 978-0-470-94994-8. https://books.google.com/books?id=ijloadAt4BQC&q=marder+condensed+matter.
- ↑ Breuer, H.-P.; Petruccione, F. (2007). The Theory of Open Quantum Systems. Oxford University Press. p. vii. ISBN 978-0-19-921390-0. ""Quantum mechanical systems must be regarded as open systems"."
Source attribution: Quantum: The Secret of Cohesion: How Waves Hold Matter Together