Physics:Quantum Partition function: Difference between revisions
imported>WikiHarold Repair Quantum Collection B backlink template |
imported>WikiHarold Repair Quantum Collection B backlink template |
(No difference)
| |
Latest revision as of 20:00, 8 May 2026
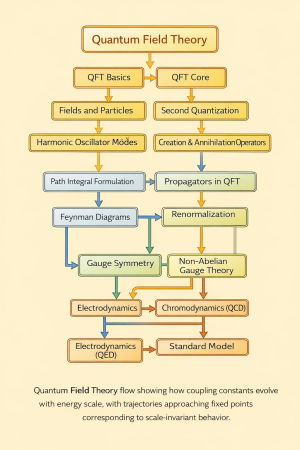
Quantum partition function is the central quantity of quantum statistical mechanics, encoding how the energy eigenstates of a quantum system are thermally populated at temperature .[1][2] It provides the bridge between microscopic quantum states and macroscopic thermodynamic quantities such as internal energy, entropy, Helmholtz free energy, and heat capacity.[3]
Definition
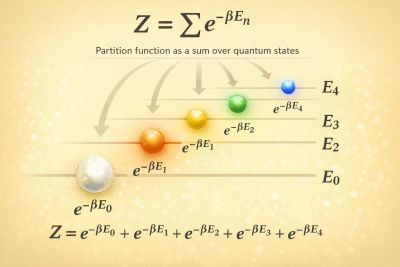
For a quantum system in thermal equilibrium with Hamiltonian , the canonical partition function is defined by
where
Here is Boltzmann’s constant, is the absolute temperature, and the trace is taken over the system’s Hilbert space.[1][4]
If the Hamiltonian has discrete energy eigenstates with eigenvalues , then the trace becomes
This shows that each energy level contributes with a Boltzmann weight determined by its energy.[2][1]
Physical meaning
The partition function summarizes the thermal accessibility of all possible quantum states of the system.[3] Low-energy states contribute most strongly at low temperature, while many higher-energy states become thermally populated as the temperature increases.[4]
Once is known, the equilibrium probability of finding the system in eigenstate is
Thus the partition function serves as the normalization factor for the canonical ensemble.[1]
Density operator
In quantum statistical mechanics, the canonical ensemble is represented by the density operator
This operator gives expectation values of observables through
The partition function therefore appears directly in the normalization of the thermal density matrix.[3][1]
Thermodynamic relations
The quantum partition function generates the main thermodynamic quantities of the canonical ensemble.[2][4]
The Helmholtz free energy is
The internal energy is
The entropy can be written as
and the heat capacity at constant volume is
These relations show that all equilibrium thermodynamics can be derived from .[1][3]
Role of degeneracy
If an energy level has degeneracy , then the partition function becomes
Degeneracy increases the statistical weight of a level and can significantly affect thermodynamic behavior, especially at low temperatures where only a few low-lying states contribute appreciably.[4]
Simple examples
Two-level system
For a system with two energy levels and , the partition function is
This model is useful for spin systems, qubits, and other simple quantum systems.[1]
Quantum harmonic oscillator
For a one-dimensional quantum harmonic oscillator with energies
the partition function is
This is one of the standard exactly solvable examples in quantum statistical mechanics.[1][4]
Ideal quantum gas
For systems of many identical particles, the partition function must reflect indistinguishability and quantum statistics.[3] In that case one passes naturally to the grand canonical formalism and to Bose-Einstein or Fermi-Dirac occupation factors.[1]
Relation to the classical partition function
The quantum partition function is the direct analogue of the classical partition function, but with the trace over Hilbert space replacing the phase-space integral.[2][3] In the semiclassical limit, where quantum level spacings become very small compared with , the quantum description approaches the classical one.[4]
This connection explains why statistical mechanics can often be formulated in a unified way, with quantum theory providing the more fundamental description.
Connection with imaginary time
In more advanced formulations, the partition function may be written as an imaginary-time evolution operator over a period :
This relation underlies the path-integral formulation of quantum statistical mechanics and connects thermal field theory with quantum dynamics in imaginary time.[5][6]
Grand partition function
When both energy and particle number may fluctuate, the appropriate quantity is the grand partition function
where is the chemical potential and is the particle-number operator.[1][3] This form is essential for describing quantum gases, photons, phonons, and many-body systems in contact with both a heat bath and a particle reservoir.
Physical interpretation
The quantum partition function explains how:
- discrete energy spectra determine thermal populations
- microscopic energy levels generate macroscopic thermodynamics
- degeneracy modifies statistical weights
- quantum statistics enters many-body equilibrium theory[1][3]
It is therefore one of the foundational objects of both quantum statistical mechanics and modern many-body physics.
Applications
Quantum partition functions are used in:
- two-level spin systems
- harmonic oscillators and lattice vibrations
- ideal Bose and Fermi gases
- magnetic systems
- quantum field theory at finite temperature
- condensed-matter and many-body physics[1][6]
See also
Table of contents (185 articles)
Index
Full contents
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 Pathria, R. K.; Beale, Paul D. (2011). Statistical Mechanics (3 ed.). Elsevier. ISBN 9780123821881.
- ↑ 2.0 2.1 2.2 2.3 Tolman, Richard C. (1979). The Principles of Statistical Mechanics. Dover Publications. ISBN 9780486638966.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 Landau, L. D.; Lifshitz, E. M. (1980). Statistical Physics, Part 1 (3 ed.). Butterworth-Heinemann. ISBN 9780750633727.
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 Reif, Frederick (2009). Fundamentals of Statistical and Thermal Physics. Waveland Press. ISBN 9781577666127.
- ↑ Feynman, Richard P.; Hibbs, Albert R. (1965). Quantum Mechanics and Path Integrals. McGraw-Hill. ISBN 9780486477220.
- ↑ 6.0 6.1 Negele, John W.; Orland, Henri (1998). Quantum Many-Particle Systems. Westview Press. ISBN 9780738200521.
Source attribution: Quantum partition function